Hansenula polymorpha (also known as Ogataea polymorpha) is a robust methylotrophic yeast expression platform widely recognized for its high secretion efficiency, thermotolerance, and ability to produce complex recombinant proteins with favorable glycosylation profiles. Compared with conventional yeast systems, H. polymorpha offers strong, tightly regulated promoters, minimal hyperglycosylation, and excellent scalability under high-cell-density fermentation conditions. These features make it a powerful alternative for producing therapeutic proteins, industrial enzymes, vaccine antigens, and challenging targets such as membrane proteins. Leveraging advanced strain engineering, fermentation optimization, and downstream processing expertise, Profacgen provides end-to-end H. polymorpha expression solutions tailored to research, diagnostic, and industrial manufacturing needs.
Yeast expression systems bridge the gap between bacterial and mammalian hosts by combining relatively low production costs with eukaryotic protein folding and post-translational modification capabilities. Among them, Hansenula polymorpha has emerged as a particularly attractive chassis due to several intrinsic biological advantages.
As a methylotrophic yeast, H. polymorpha can utilize methanol as a carbon source, enabling the use of strong, tightly regulated inducible promoters such as MOX (methanol oxidase) and FMD (formate dehydrogenase). These promoters allow precise control of gene expression and support exceptionally high protein yields under optimized fermentation conditions.
Unlike Saccharomyces cerevisiae, which often retains recombinant proteins intracellularly, H. polymorpha demonstrates high secretion efficiency, allowing heterologous proteins to be released directly into the culture medium. This significantly simplifies downstream purification and reduces overall production costs.
Another key advantage is its thermotolerance. H. polymorpha grows well at temperatures up to 45–50 °C, enabling faster fermentation cycles, reduced contamination risk, and improved process robustness—attributes that are highly valued in large-scale industrial settings.
Importantly, H. polymorpha exhibits minimal hyperglycosylation compared with Pichia pastoris, producing glycoproteins with shorter, more homogeneous mannose chains. This characteristic lowers immunogenicity risk and improves suitability for therapeutic and diagnostic applications.
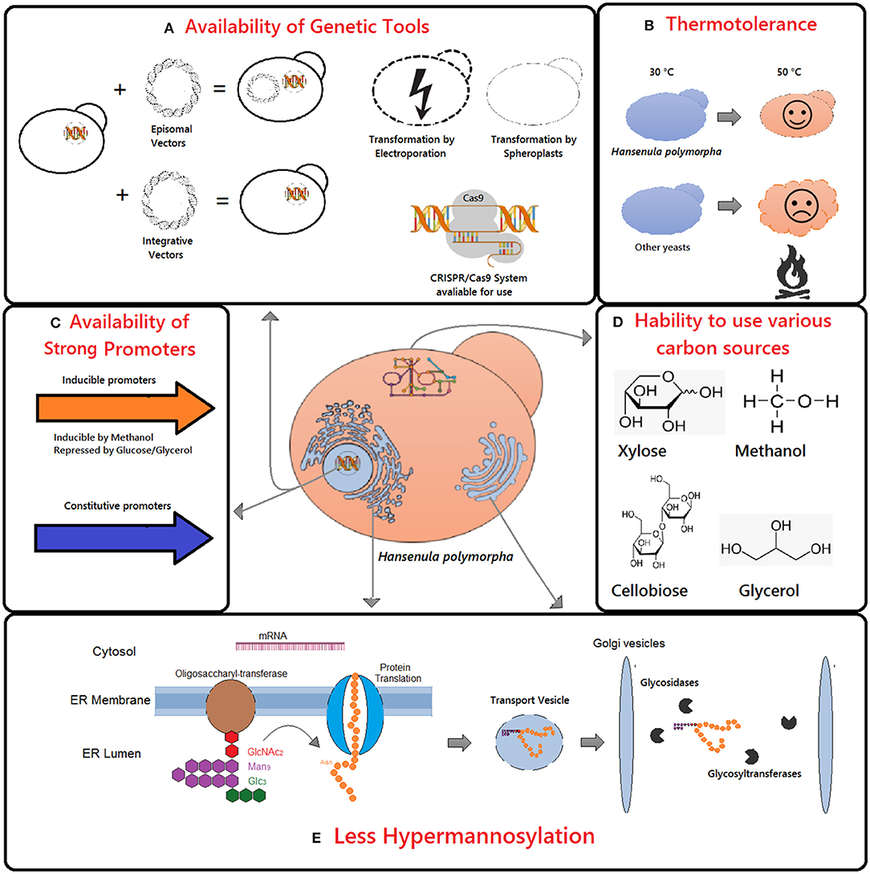 Figure 1. Main advantages of Hansenula polymorpha as chassis for recombinant protein production include the availability of genetic tools (A,C), thermotolerance (B), ability to use various carbon sources (D), and glycosylation pattern (E). (Manfrão-Netto et al., 2019)
Figure 1. Main advantages of Hansenula polymorpha as chassis for recombinant protein production include the availability of genetic tools (A,C), thermotolerance (B), ability to use various carbon sources (D), and glycosylation pattern (E). (Manfrão-Netto et al., 2019)
We provide end-to-end recombinant protein expression services using Hansenula polymorpha, from gene design to purified protein delivery. Our workflows are modular and fully customizable to meet diverse application requirements.
Strain Engineering and Vector Design
We design and construct expression strains optimized for yield, secretion, and product quality:
Protein Expression and Fermentation Optimization
Our fermentation strategies are developed to maximize productivity and reproducibility:
Downstream Processing and Purification
To deliver highly pure, functional proteins, we provide integrated purification workflows:
Analytical Characterization and Quality Control
Comprehensive analytical services ensure identity, purity, and bioactivity:
Thanks to its secretion efficiency, scalability, and favorable glycosylation behavior, H. polymorpha is widely applied across multiple sectors.
Background
A biotechnology company required gram-scale production of a secreted therapeutic cytokine for preclinical pharmacology and formulation studies. The target protein required correct folding, disulfide bond formation, and consistent bioactivity, while minimizing downstream purification complexity and development timelines.
Our Solution
We engineered a multi-copy Hansenula polymorpha expression strain using the methanol-inducible MOX promoter combined with systematic signal peptide screening to enhance secretion efficiency. Fed-batch fermentation parameters, including induction strategy, temperature, and feed rate, were optimized to support high-cell-density growth and stable expression.
Final Results
The cytokine was efficiently secreted into the culture supernatant at high titers with correct molecular weight and bioactivity. Final purity exceeded 95% after streamlined purification, enabling rapid advancement into downstream preclinical studies.
Background
An industrial enzyme manufacturer sought a cost-effective and scalable expression system to produce a thermostable enzyme for detergent formulations. Key requirements included high volumetric yield, operational robustness, and enzyme stability under harsh processing and storage conditions.
Our Solution
A thermotolerant H. polymorpha strain was developed, leveraging its ability to grow at elevated temperatures. Expression was driven by a strong inducible promoter, and high-cell-density fed-batch fermentation was optimized to maximize productivity. Process conditions were tailored to maintain enzyme integrity while supporting large-scale scalability.
Final Results
The enzyme was produced at high yield with excellent thermal and functional stability. The optimized process significantly reduced production costs and demonstrated reliable performance during scale-up, supporting the client's transition to commercial manufacturing.
Background
A vaccine developer required recombinant production of a viral surface antigen with minimal hyperglycosylation and consistent glycan profiles. The antigen needed to maintain structural integrity and immunogenicity to support downstream vaccine formulation and evaluation.
Our Solution
We selected H. polymorpha for its reduced hypermannosylation compared with other yeast systems and engineered an expression strain with optimized secretion signals and controlled induction conditions. Fermentation and expression parameters were fine-tuned to balance yield, secretion efficiency, and glycosylation consistency.
Final Results
The antigen was secreted efficiently with uniform glycan patterns and strong immunogenic properties. Analytical characterization confirmed batch-to-batch consistency, meeting quality expectations for vaccine development and enabling progression toward further preclinical studies.
Consult Our Experts on Your Project
With its superior secretion capacity, thermotolerance, and favorable glycosylation behavior, Hansenula polymorpha represents a powerful and versatile yeast expression platform. Our integrated expertise in strain engineering, fermentation optimization, and downstream processing ensures reliable delivery of high-quality recombinant proteins for research, diagnostic, and therapeutic applications. Partner with us to fully leverage the strengths of H. polymorpha for your next protein production project.
Reference:
Fill out this form and one of our experts will respond to you within one business day.