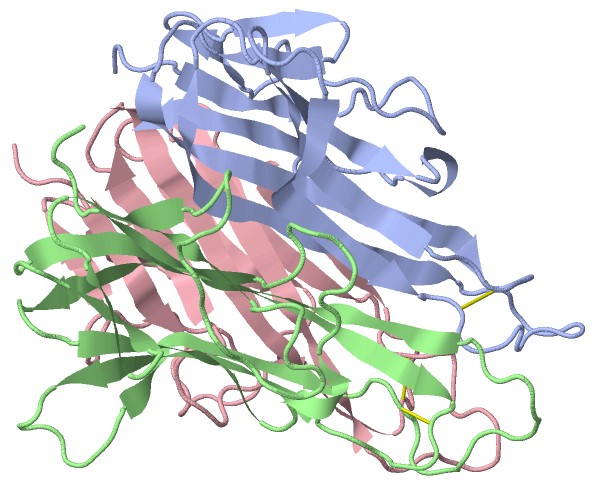
 Figure 1. Trimeric structure of TNF alpha.
Figure 1. Trimeric structure of TNF alpha.
Protein trimerization refers to the assembly of three identical or non-identical protein subunits into a stable macromolecular complex, typically through noncovalent interactions or engineered covalent linkages. Trimeric proteins play critical roles in biological systems, particularly in signaling pathways, immune responses, and viral entry mechanisms.
At Profacgen, we provide advanced trimeric protein production services based on proprietary design and expression strategies that enable the generation of disulfide bond-linked homo-trimeric proteins with enhanced stability, bioactivity, and pharmacokinetic performance. Our platform supports efficient expression, scalable purification, and rigorous characterization of trimeric proteins for applications in therapeutic development, vaccine design, and structural biology. With extensive experience and optimized workflows, we deliver high-quality trimeric proteins from early research stages through preclinical development.
Protein trimerization is defined as the formation of a trimeric complex consisting of three subunits that associate through noncovalent interactions or covalent linkages. These subunits may be identical (homotrimers) or different (heterotrimers), forming a structurally and functionally distinct assembly.
Trimeric protein structures are widely found in nature and are particularly important in:
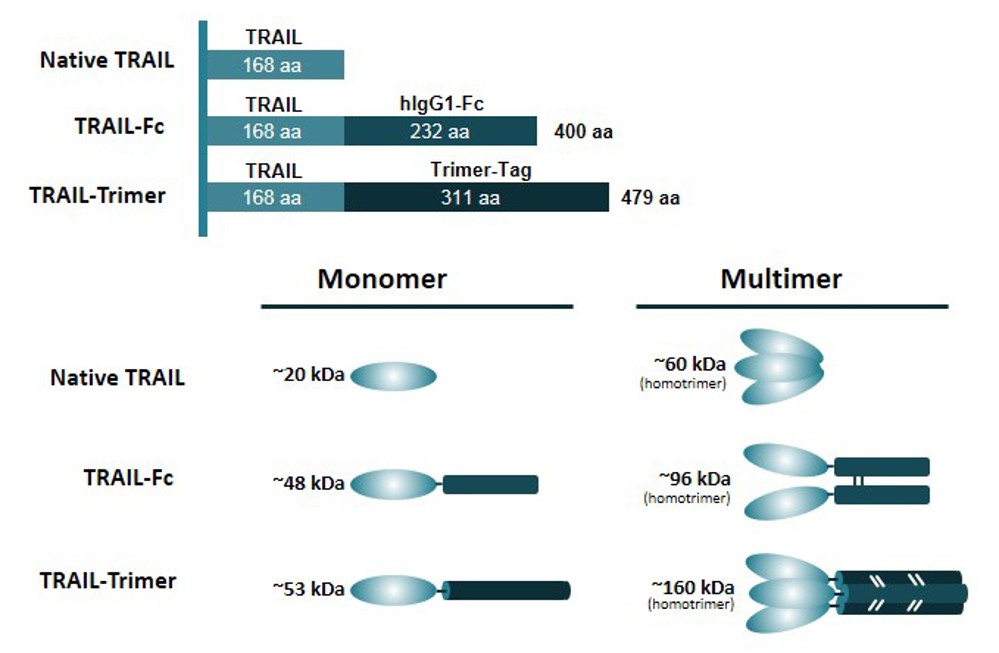 Figure 2. Example of trimeric proteins: different TRAIL polypeptides. (Liu et al., 2017)
Figure 2. Example of trimeric proteins: different TRAIL polypeptides. (Liu et al., 2017)
The biological activity of many proteins depends on their oligomeric state. In particular, trimeric structures provide:
Currently, many biologic drugs targeting trimeric ligands—such as TNF inhibitors—are designed as dimeric molecules. However, from a structural biology perspective, a dimer with two-fold symmetry cannot perfectly interact with a trimeric target possessing three-fold symmetry. This mismatch can lead to:
To overcome these limitations, engineered trimeric proteins have been developed to better match the natural structure of their targets. By designing covalently linked homotrimers, researchers can achieve:
Trimeric protein engineering has shown significant promise in multiple therapeutic areas, including autoimmune diseases, cancer, infectious diseases, osteoporosis, and cardiovascular disorders.
Profacgen provides a comprehensive and advanced trimeric protein production platform, integrating rational design, expression optimization, and downstream characterization to ensure successful generation of high-quality trimeric proteins.

Our trimerization platform enables the production of proteins with the following features:
This platform establishes a broadly applicable system for the production and characterization of trimeric proteins.
Our trimeric protein production service supports diverse applications:
Many of these constructs are under development as biologic drugs.
Client Requirements:
A biotech company aimed to develop a therapeutic protein targeting a trimeric cytokine involved in tumor progression. Monomeric and dimeric receptor candidates showed insufficient binding avidity and limited antitumor activity in preliminary studies.
Our Solution:
Profacgen designed a disulfide bond-linked homotrimeric receptor construct engineered to precisely match the three-fold symmetry of the native target cytokine. The construct was expressed and optimized in a mammalian system to ensure proper folding, assembly, and post-translational modifications essential for receptor function.
Final Results:
The resulting trimeric protein demonstrated significantly enhanced binding affinity to the cytokine target compared to dimeric counterparts, driven by avidity effects. In vitro antitumor activity assays revealed superior functional potency, with enhanced receptor signaling and tumor cell growth inhibition. The client advanced this lead candidate into preclinical development.
Client Requirements:
A research group needed a stable trimeric form of a viral envelope protein for vaccine immunogenicity studies. Native-like trimeric conformation was essential to elicit neutralizing antibodies, but previous monomeric preparations failed to induce protective responses.
Our Solution:
We engineered a trimeric fusion construct incorporating a foldon trimerization domain to mimic the native prefusion structure of the viral protein. Signal peptide optimization and expression screening in mammalian cells maximized secretion of correctly assembled trimeric antigen.
Final Results:
Successfully produced a structurally stable trimeric antigen with conformation confirmed by negative-stain electron microscopy and antibody binding studies. Immunogenicity studies demonstrated significantly enhanced neutralizing antibody responses compared to monomeric controls, enabling effective evaluation in the client's vaccine development pipeline.
Client Requirements:
The client sought to improve the half-life and stability of a therapeutic protein that showed promising bioactivity but suffered from rapid clearance and poor stability in vivo.
Our Solution:
Profacgen applied its proprietary trimerization platform to generate a covalently linked trimeric version of the protein using engineered disulfide bonds at the C-terminus. Expression and purification conditions were systematically optimized to maximize yield of correctly assembled trimer while minimizing aggregate formation.
Final Results:
The trimeric protein exhibited significantly improved pharmacokinetic properties with extended circulation half-life compared to the monomeric form. Stability studies confirmed enhanced resistance to thermal and proteolytic degradation. The optimized trimeric candidate demonstrated strong potential for therapeutic applications, supporting the client's advancement toward preclinical studies.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.