Bacterial protein expression remains the most widely adopted and cost-effective platform for the production of recombinant proteins across basic research, biotechnology, diagnostics, and early-stage biopharmaceutical development. Leveraging rapid host growth, simple culture requirements, and exceptional scalability, bacterial systems enable fast turnaround and high-yield protein production at both laboratory and industrial scales. Profacgen provides comprehensive bacterial protein expression services centered on Escherichia coli and advanced Gram-positive bacterial hosts, complemented by specialized systems including anaerobic bacteria, lactic acid bacteria, and Corynebacterium glutamicum. Through systematic feasibility evaluation, rational construct design, and robust process optimization, we deliver high-quality recombinant proteins tailored to diverse downstream applications.
Bacterial hosts were the first systems successfully applied to recombinant protein expression and continue to serve as the backbone of modern protein production. Their enduring popularity stems from several intrinsic advantages: rapid doubling times, well-characterized genetics, straightforward culture conditions, and ease of scale-up from milliliter cultures to industrial fermenters.
Among bacterial hosts, E. coli is the most extensively studied and widely used expression system. Its well-understood molecular biology, extensive strain library, and rich toolbox of promoters, fusion tags, and expression vectors make it the default first-choice host for many recombinant protein projects. E. coli has been successfully used to produce a wide range of recombinant proteins, including enzymes, cytokines, antigens, immunogens, antibody fragments, and protein domains.
In parallel, Gram-positive bacteria such as Bacillus subtilis have gained increasing attention, particularly for applications requiring secreted proteins or low endotoxin levels. As an FDA-designated Generally Recognized as Safe (GRAS) organism, B. subtilis offers regulatory advantages and eliminates concerns associated with endotoxin contamination. Its natural secretion pathways allow recombinant proteins to be released directly into the culture medium, greatly simplifying downstream purification. Additionally, B. subtilis exhibits reduced codon bias compared to E. coli, facilitating the expression of genes from diverse origins.
Despite these advantages, bacterial expression is not without challenges. Proteins may form inclusion bodies, lack post-translational modifications, or exhibit improper folding. Certain targets—such as toxic proteins, oxygen-sensitive enzymes, or proteins requiring secretion—may not be well suited to conventional E. coli systems. Addressing these challenges requires not only system selection but also careful engineering and optimization across the entire expression workflow. Profacgen's bacterial expression services are designed to overcome these limitations through flexible host selection, advanced construct design, and integrated upstream and downstream optimization.
Profacgen offers a comprehensive and customizable portfolio of bacterial protein expression services, covering standard and specialized bacterial systems to meet diverse technical and application-driven requirements.
Escherichia coli Protein Expression
Bacillus subtilis Expression System
Anaerobic Bacteria Protein Expression Service
Lactic Acid Bacteria (LAB) Protein Expression Service

Corynebacterium glutamicum Expression System
Across all bacterial systems, Profacgen supports both intracellular and secretory expression strategies, enabling rational selection of hosts and workflows based on protein properties and downstream use.
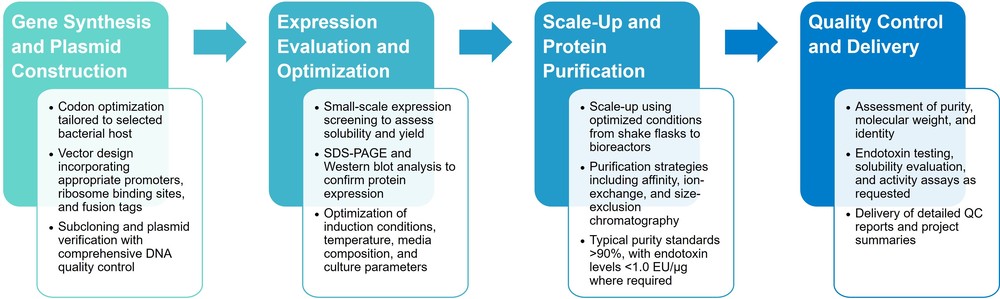
Our bacterial protein expression services follow a structured, data-driven workflow designed to maximize expression success while minimizing project risk.
Discuss Your Manufacturing Timeline
Protein Labeling
Endotoxin Detection and Removal
Inclusion Body Solubilization and Refolding
Background
A client required gram-scale production of a recombinant enzyme for high-throughput screening. Previous attempts resulted in insoluble inclusion bodies, which compromised catalytic activity and stalled lead optimization.
Our Solution
We implemented a "solubility-first" strategy, beginning with codon optimization and the screening of specialized host strains and fusion tags. To prevent aggregation, we optimized induction conditions by lowering temperatures and using auto-induction media to favor proper folding. High-homogeneity was ensured through a multi-step purification process.
Final Results
The enzyme was delivered with >95% purity and high catalytic activity. This successful soluble expression provided the gram-scale quantities needed for the client's screening campaign, significantly accelerating their drug discovery timeline.
Background
The client needed a soluble protein for sensitive cell-based assays. Conventional E. coli systems raised concerns regarding endotoxin (LPS) contamination, which triggers non-specific immune responses and yields unreliable assay data.
Our Solution
We utilized a Bacillus subtilis secretion system, a Gram-positive platform that naturally lacks endotoxins. By screening signal peptide libraries, we optimized the Sec-pathway translocation, allowing the target protein to be secreted directly into the medium. This eliminated the need for complex endotoxin removal steps.
Final Results
The protein was delivered with endotoxin levels <1.0 EU/μg and retained full biological activity. This streamlined, endotoxin-free approach provided the client with high-purity material, ensuring reproducible and accurate results in their functional studies.
Background
A client required the functional expression of an oxygen-sensitive oxidoreductase. The enzyme's metal-sulfur clusters degraded rapidly upon atmospheric exposure, leading to a total loss of activity in standard aerobic systems.
Our Solution
We deployed a specialized anaerobic platform. The entire process—from fermentation to harvesting—was conducted in a strictly oxygen-free environment. We used degassed buffers and specialized reducing agents during purification to stabilize the enzyme's delicate active site and prevent oxidative damage.
Final Results
The purified enzyme retained its native catalytic activity and structural integrity. Our rigorous anaerobic handling enabled the client to perform successful biochemical characterization, supporting their metabolic engineering and pathway optimization efforts.
Profacgen has accumulated extensive experience in bacterial protein expression and related technologies. Our professional technical team delivers high-quality recombinant proteins through reliable systems, competitive pricing, and responsive project management. Contact us to discuss how our bacterial expression services can accelerate your research and development programs.
Fill out this form and one of our experts will respond to you within one business day.