Unnatural amino acid (UAA) technology represents a cutting-edge protein engineering strategy that enables site-specific modification of therapeutic proteins to enhance pharmacokinetic properties, including half-life, stability, and bioavailability. By expanding the genetic code through engineered tRNA/synthetase systems, UAAs with unique chemical functionalities can be incorporated into proteins at defined positions, enabling precise and controlled molecular design.
Unlike traditional modification approaches, UAA technology allows homogeneous, site-selective conjugation of half-life extending moieties such as polymers, lipids, or functional groups, minimizing disruption to protein structure and activity. This precision makes UAA-based engineering particularly valuable for next-generation biologics and biobetters.
At Profacgen, we provide comprehensive UAA-enabled protein engineering services, including expression system development, site-specific incorporation, conjugation strategies, and functional validation. Our platform supports the development of highly optimized long-acting therapeutics across diverse applications.
Protein and peptide therapeutics have transformed modern medicine, yet their clinical utility is often limited by rapid clearance, enzymatic degradation, and suboptimal pharmacokinetics. Traditional strategies for extending half-life—such as PEGylation, Fc fusion, and albumin fusion—have achieved considerable success but are often constrained by limitations in control, homogeneity, and structural flexibility.
Unnatural amino acid technology provides a fundamentally different and highly precise approach to protein modification. Rather than modifying proteins randomly or at multiple sites, UAA incorporation allows the introduction of non-canonical amino acids at predefined positions, enabling controlled functionalization.
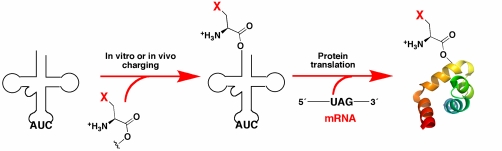 Figure 1. Unnatural amino acid incorporation though nonsense codon suppression. (Minnihan et al., 2009)
Figure 1. Unnatural amino acid incorporation though nonsense codon suppression. (Minnihan et al., 2009)
Unnatural amino acids are amino acids that are not among the 20 standard amino acids encoded by the natural genetic code. These molecules can be designed to include unique chemical functionalities, such as:
These properties allow proteins to be engineered with enhanced stability, reduced degradation, and improved pharmacokinetic behavior.
The incorporation of UAAs relies on genetic code expansion, typically achieved through:
This system enables site-specific insertion of UAAs during translation, ensuring precise and reproducible modification.
UAA technology contributes to half-life extension through several mechanisms:
For example, lipidation of peptide drugs (e.g., GLP-1 analogs) significantly increases binding to serum proteins, thereby extending circulation time.
Cell-free protein synthesis systems provide an efficient platform for UAA incorporation, as they allow direct addition of unnatural amino acid substrates into the reaction mixture. This enables:
In addition to CFPS, UAA incorporation can also be achieved in prokaryotic and mammalian expression systems, expanding its applicability across different protein types.
Profacgen provides a comprehensive UAA-enabled protein engineering platform, supporting the development of optimized long-acting therapeutics through precise molecular design and advanced expression technologies.

Genetic Code Expansion in Cell-Based Systems
We provide UAA incorporation services in multiple expression hosts:
Capabilities include:
Cell-Free Protein Synthesis (CFPS) Platform
Our CFPS platform enables rapid and flexible UAA incorporation:
This approach is particularly valuable for early-stage discovery and optimization.
UAA incorporation enables precise attachment of functional moieties:
Depending on your molecule type and therapeutic goals, alternative or complementary strategies may provide additional advantages:
Client Requirements:
A peptide therapeutic candidate exhibited rapid renal clearance and susceptibility to proteolytic degradation, resulting in poor pharmacokinetics and frequent dosing requirements that limited its clinical potential.
Our Solution:
An unnatural amino acid (UAA) containing a reactive functional group was site-specifically incorporated at a defined position distal to the peptide's active site. This enabled controlled, stoichiometric conjugation with a half-life extending moiety, ensuring uniform product without random off-target modification.
Final Results:
The modified peptide demonstrated significantly improved pharmacokinetics with circulation time extended more than tenfold, while biological activity was fully retained as confirmed by receptor-binding assays. The site-specific approach ensured product homogeneity, supporting the client's advancement into preclinical development with a well-characterized candidate.
Client Requirements:
A recombinant protein therapeutic suffered from rapid in vivo degradation by serum proteases, leading to poor stability and suboptimal exposure that compromised efficacy.
Our Solution:
Strategic incorporation of D-type unnatural amino acids at protease-susceptible cleavage sites was performed while preserving regions critical for biological activity. Multiple variants were designed and screened to identify the optimal balance between protease resistance and functional integrity.
Final Results:
The engineered protein demonstrated dramatically enhanced stability in serum stability assays with significantly reduced proteolytic cleavage. Half-life was extended more than fivefold in pharmacokinetic studies, while functional assays confirmed fully maintained activity. The client obtained a stabilized candidate suitable for further development without compromising therapeutic efficacy.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.