Recombinant protein expression is a cornerstone technology in modern life sciences and biopharmaceutical development. By leveraging recombinant DNA technology, target proteins are produced efficiently in carefully selected host systems to meet specific research, diagnostic, or therapeutic needs. Profacgen provides comprehensive, customized protein expression services covering multiple expression platforms, from bacterial and yeast systems to mammalian, insect, cell-free, and animal-free expression. Through rigorous feasibility analysis, advanced engineering strategies, and end-to-end project management, we ensure high-yield, high-quality protein production suitable for functional assays, structural studies, target validation, and industrial-scale applications.
Recombinant protein expression involves introducing a target gene—typically a cDNA—into an appropriate host cell under the control of a well-characterized promoter, enabling controlled and high-level protein production. This technology is now indispensable in basic research, drug discovery, and the manufacture of recombinant protein therapeutics.
However, protein expression is rarely a one-size-fits-all process. Protein size, solubility, subcellular localization, post-translational modifications (PTMs), folding requirements, and intended downstream applications all critically influence system selection and process design. Complex targets such as membrane proteins, large multi-domain proteins, nuclear proteins, or heavily modified eukaryotic proteins often require advanced strategies and iterative optimization. To address these challenges, Profacgen assembles experienced protein biologists to conduct thorough feasibility evaluations prior to project initiation, minimizing risk and maximizing success rates.
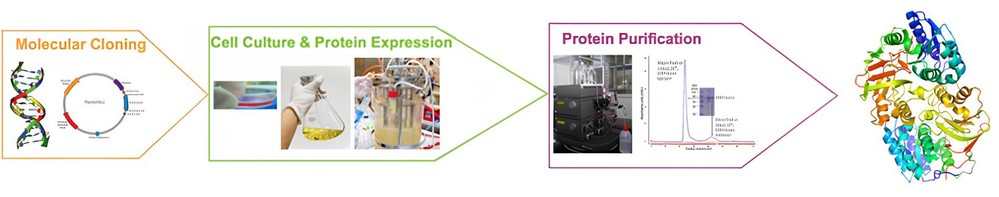
Our protein expression services cover the complete workflow from gene to purified protein, as depicted in the standard recombinant protein production pipeline. Starting with molecular cloning and expression vector construction, we proceed through optimized cell culture and protein expression in selected host systems, followed by scalable protein purification using advanced chromatographic platforms. By integrating upstream expression strategies with downstream purification development, we ensure that the final protein product meets stringent requirements for purity, structural integrity, and biological activity, supporting applications ranging from functional assays and structural biology to therapeutic research and industrial use.
Profacgen offers a full portfolio of recombinant protein expression platforms. These platforms have flexible customization and advanced optimization options.
Insect Cell / Baculovirus Expression
Membrane Protein Expression and Purification
Specific and Customized Expression Strategies
High-Throughput Engineering of Signal Peptides
Across all platforms, Profacgen provides end-to-end services including gene synthesis, codon optimization, vector construction, host cell culture, protein expression, purification, and rigorous quality control.
Background
The client required milligram-to-gram quantities of an active recombinant enzyme for high-throughput biochemical screening. Key challenges included low predicted solubility and the need for consistent batch-to-batch activity.
Our Solution
We selected a bacterial expression system combined with gene synthesis and codon optimization. Multiple fusion tags and expression strains were screened in parallel to enhance solubility. Expression conditions, including induction temperature and timing, were systematically optimized, followed by streamlined affinity-based purification.
Final Results
High-level soluble expression was achieved, delivering >95% pure enzyme with robust catalytic activity. The final yield fully supported the client's screening campaign and enabled rapid progression of their discovery program.
Background
The client sought a eukaryotic glycoprotein with native folding and authentic post-translational modifications for cell-based functional assays. Transient expression alone did not provide sufficient consistency for extended studies.
Our Solution
A mammalian cell expression strategy was implemented using HEK/CHO systems. After feasibility assessment, we constructed a stable cell line to ensure long-term, reproducible protein production. Signal peptide selection and secretion conditions were optimized, followed by multi-step chromatographic purification.
Final Results
The purified protein displayed correct molecular weight, glycosylation patterns, and high biological activity. Stable production ensured reliable supply for ongoing functional assays and downstream validation studies.
Background
The client required a structurally intact membrane protein suitable for cryo-EM analysis. Major challenges included low expression levels and protein instability during solubilization.
Our Solution
We employed a baculovirus–insect cell expression system, combined with customized construct design and expression optimization. Detergent screening and purification condition development were conducted in parallel to stabilize the target protein and preserve native conformation.
Final Results
The final product met purity and homogeneity requirements for cryo-EM studies, enabling high-resolution structural analysis. The client successfully advanced their structure-based drug discovery program.
Consult Our Experts on Your Project
Fill out this form and one of our experts will respond to you within one business day.