Fusion proteins are powerful tools for enhancing protein expression, purification, stability, and therapeutic performance. By genetically linking functional domains—such as carrier proteins, Fc fragments, or targeting ligands—fusion constructs enable improved pharmacokinetics, bioavailability, and biological activity.
At Profacgen, we offer a comprehensive, end-to-end fusion protein expression platform, covering rational design, cloning, expression, purification, and functional validation. Our integrated capabilities support both research-grade and therapeutic development projects, ensuring high-quality protein production with optimized yield and functionality.
Fusion proteins are widely used to investigate protein functions and to facilitate protein expression and purification, as well as to promote protein bioavailability. In biological medicine, fusion proteins play particularly important roles in prolonging in vivo half-life and enabling targeted delivery of drug candidates.
One of the most promising fusion protein strategies involves Human Serum Albumin (HSA). Over the past decades, albumin has been extensively studied as a drug delivery vector, especially in tumor treatment, due to its natural accumulation in solid tumors.
Because of this property, gene fusion with albumin has attracted increasing attention in the pharmaceutical industry. Representative examples include:
To date, more than 60 recombinant HSA-fusion therapeutic proteins have been successfully expressed, with several advancing into clinical stages.
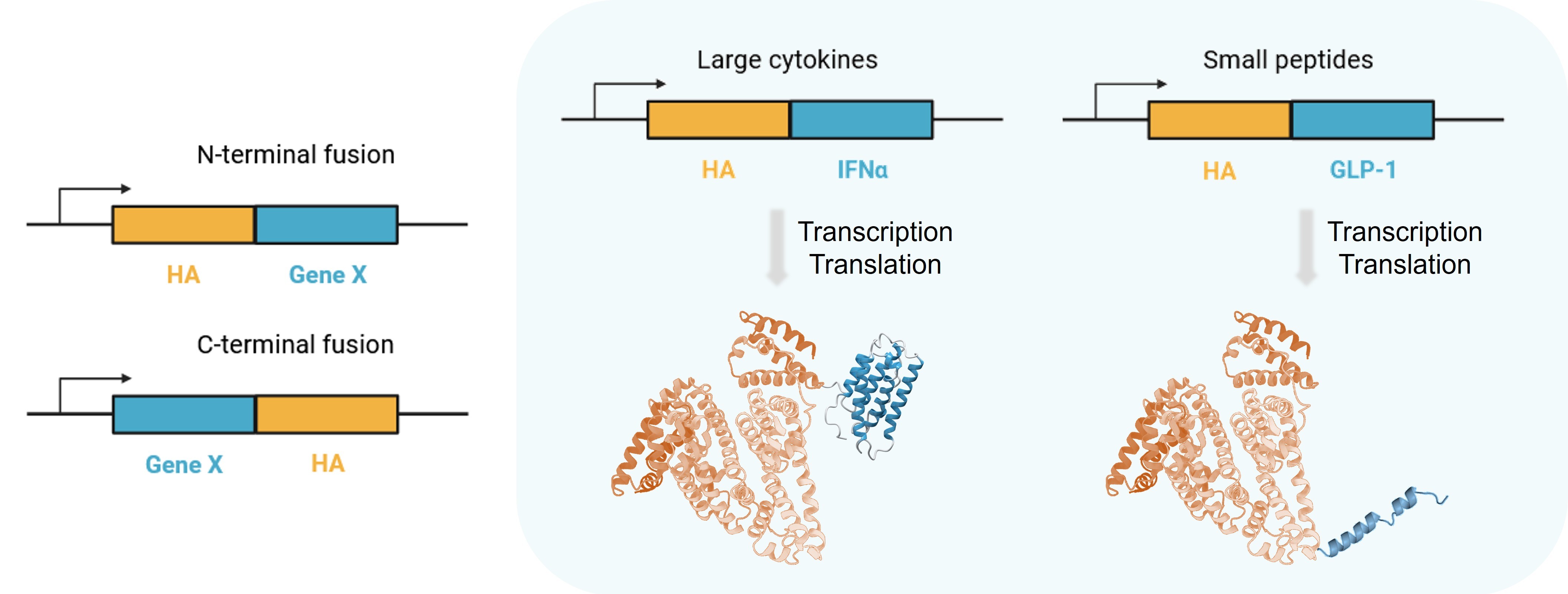 Figure 1. Human serum albumin (HA) fused therapeutic protein.
Figure 1. Human serum albumin (HA) fused therapeutic protein.
Another widely adopted approach is Fc fusion. Many proteins suffer from short plasma half-life; however, fusion with the Fc fragment of immunoglobulins can significantly extend circulation time and reduce immunogenicity.
Successful Fc-fusion therapeutics include:
Recent studies also show that mutations at the Fc dimerization interface can further enhance protein stability, offering new opportunities for therapeutic optimization. In addition to proteins, small molecules can also be conjugated to Fc fragments to improve stability.
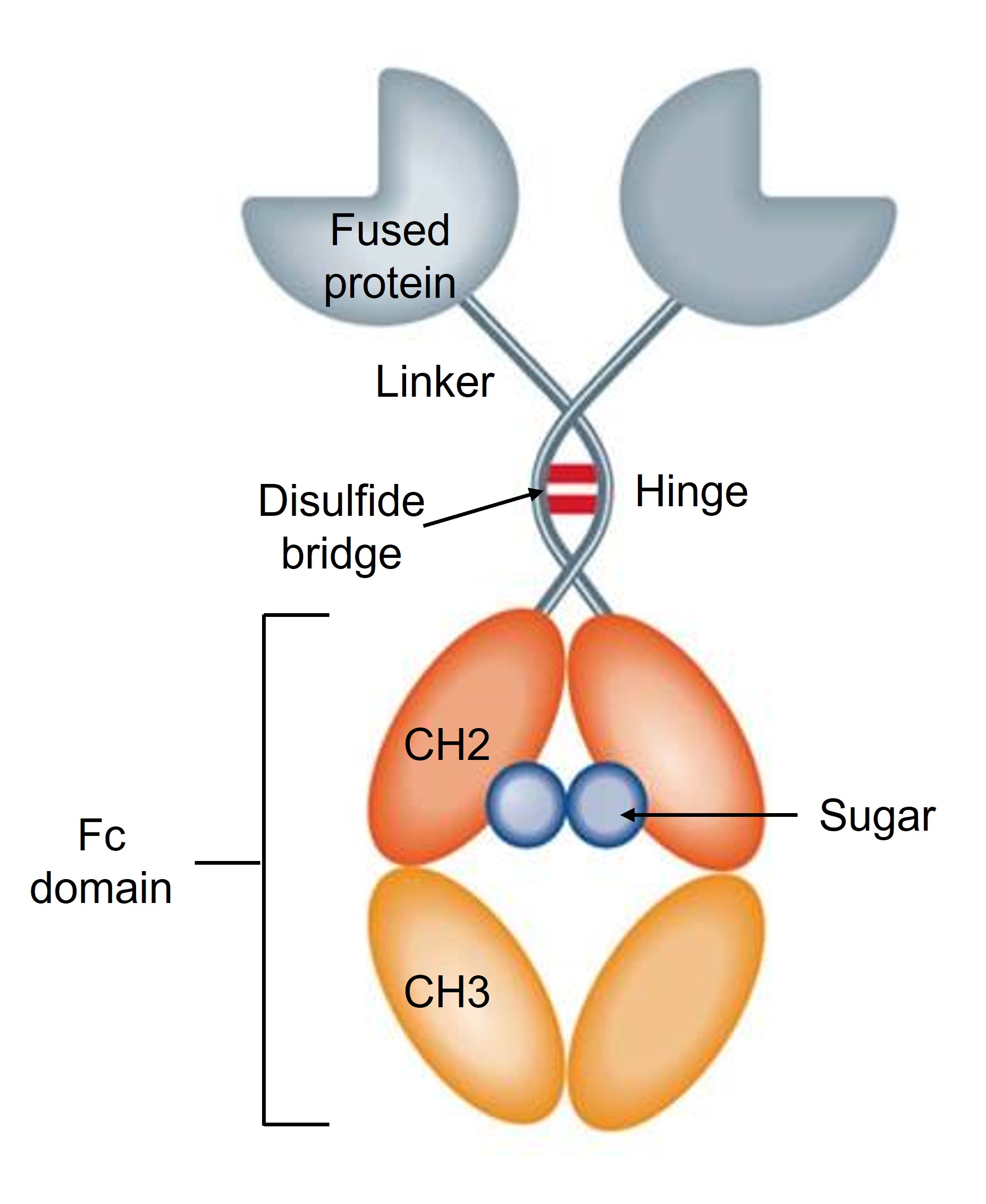 Figure 2. Fc-fusion protein.
Figure 2. Fc-fusion protein.
We provide a one-stop fusion protein production service, spanning from gene design to functional validation. Our platform integrates advanced molecular biology, protein engineering, and expression technologies.
Fusion Construct Design
Gene Synthesis & Cloning
Expression System Selection
We support both cell-based and cell-free systems:
Expression System Optimization
Process optimization includes:
Protein Purification
Protein Characterization & Quality Control
To better meet diverse research and therapeutic needs, we offer specialized fusion protein services, each tailored for specific fusion strategies and applications. Explore the modules below to access detailed service pages:
Fc Fusion Protein Production Service
Fc fusion enhances protein half-life, stability, and reduces immunogenicity, making it ideal for therapeutic candidates. Our service covers design, expression, purification, and functional validation of Fc-fusion proteins to support research and clinical development.
HSA Fusion Protein Production Service
Human Serum Albumin (HSA) fusion is widely used to prolong circulation time and enable targeted delivery. We provide comprehensive HSA-fusion services, including construct optimization, expression in multiple systems, and activity assessment, ensuring high-quality protein for preclinical or research applications.
Serum IgG fusion allows for extended half-life and functional enhancement of protein therapeutics. This service module includes gene design, cloning, expression, purification, and characterization, providing reliable solutions for advanced therapeutic protein development.
Client Requirements:
A biotechnology company had developed a promising peptide therapeutic, but its clinical potential was severely limited by rapid clearance from circulation, which would necessitate impractical frequent dosing. The client needed a strategy to significantly prolong the peptide's half-life without compromising its inherent biological activity.
Our Solution:
We proposed fusing the therapeutic peptide to Human Serum Albumin (HSA), a natural carrier with long half-life. Our team engineered several fusion constructs with different flexible linkers to ensure proper independent folding of both moieties. The optimal construct was expressed in a stable CHO cell system to guarantee correct mammalian post-translational modifications.
Final Results:
The purified HSA-fusion protein demonstrated significantly improved stability in vitro with marked resistance to proteolytic degradation. Surface plasmon resonance confirmed retained binding affinity to the target. Pharmacokinetic studies revealed a dramatically extended half-life from minutes to over 20 hours, validating the fusion strategy and advancing the client's program to the next stage.
Client Requirements:
A client working on a novel recombinant cytokine faced significant challenges with protein instability. The cytokine was prone to aggregation and rapid degradation during storage, which also led to inconsistent and reduced bioactivity. The client needed a robust solution to stabilize the protein while maintaining its therapeutic efficacy.
Our Solution:
We recommended engineering an Fc-fusion protein by linking the cytokine to the Fc region of a human antibody. This strategy leverages the Fc domain's ability to enhance solubility and stability through its well-characterized structure. The construct was designed with a short linker to minimize interference with receptor interaction and transfected into CHO cells for stable expression.
Final Results:
The expressed Fc-cytokine fusion protein exhibited superior stability with significantly reduced aggregation as measured by dynamic light scattering. The fusion partner also facilitated a simple two-step purification process via Protein A chromatography. Bioactivity assays revealed that the stabilized cytokine not only retained but demonstrated enhanced potency compared to the unstable native form.
Consult Our Experts on Your Project
Fill out this form and one of our experts will respond to you within one business day.