XTENylation is an advanced half-life extension strategy that utilizes genetically encoded, unstructured polypeptide polymers to enhance the pharmacokinetic properties of therapeutic proteins and peptides. XTEN is a hydrophilic, biodegradable protein polymer characterized by a large hydrodynamic volume and negative charge, enabling it to significantly reduce renal clearance and improve molecular stability.
By fusing XTEN sequences to biologically active molecules, this technology enhances solubility, prolongs plasma half-life, and reduces aggregation without introducing non-biological components. Compared to traditional approaches such as PEGylation, XTENylation offers improved biosafety, homogeneity, and tunability.
At Profacgen, we provide comprehensive XTENylation services, including molecular design, recombinant expression, chemical conjugation, and functional validation. Our platform supports the development of long-acting biologics across a wide range of therapeutic areas, including hormones, enzymes, coagulation factors, and peptide drugs.
The clinical success of protein- and peptide-based therapeutics has been accompanied by persistent challenges related to their pharmacokinetic limitations. Many biologics are rapidly cleared from circulation due to their relatively small size, susceptibility to enzymatic degradation, and interactions with clearance pathways. These limitations often necessitate frequent dosing, which can negatively impact patient compliance and increase treatment costs.
To overcome these challenges, a variety of half-life extension technologies have been developed. Among them, polymer-based strategies have gained significant attention for their ability to increase the effective size and stability of therapeutic molecules. Traditional approaches such as PEGylation have demonstrated success but are associated with several drawbacks, including potential immunogenicity, non-biodegradability, and heterogeneous conjugation profiles.
XTENylation represents a next-generation alternative that addresses many of these limitations. XTEN is a genetically encoded, unstructured polypeptide polymer composed of non-repetitive sequences of six hydrophilic and chemically stable amino acids. This design confers several unique properties:
Unlike synthetic polymers, XTEN is produced through recombinant expression, ensuring uniformity and reproducibility. This enables the generation of homogeneous fusion proteins with predictable pharmacokinetic behavior.
XTENylation can be implemented through two primary strategies:
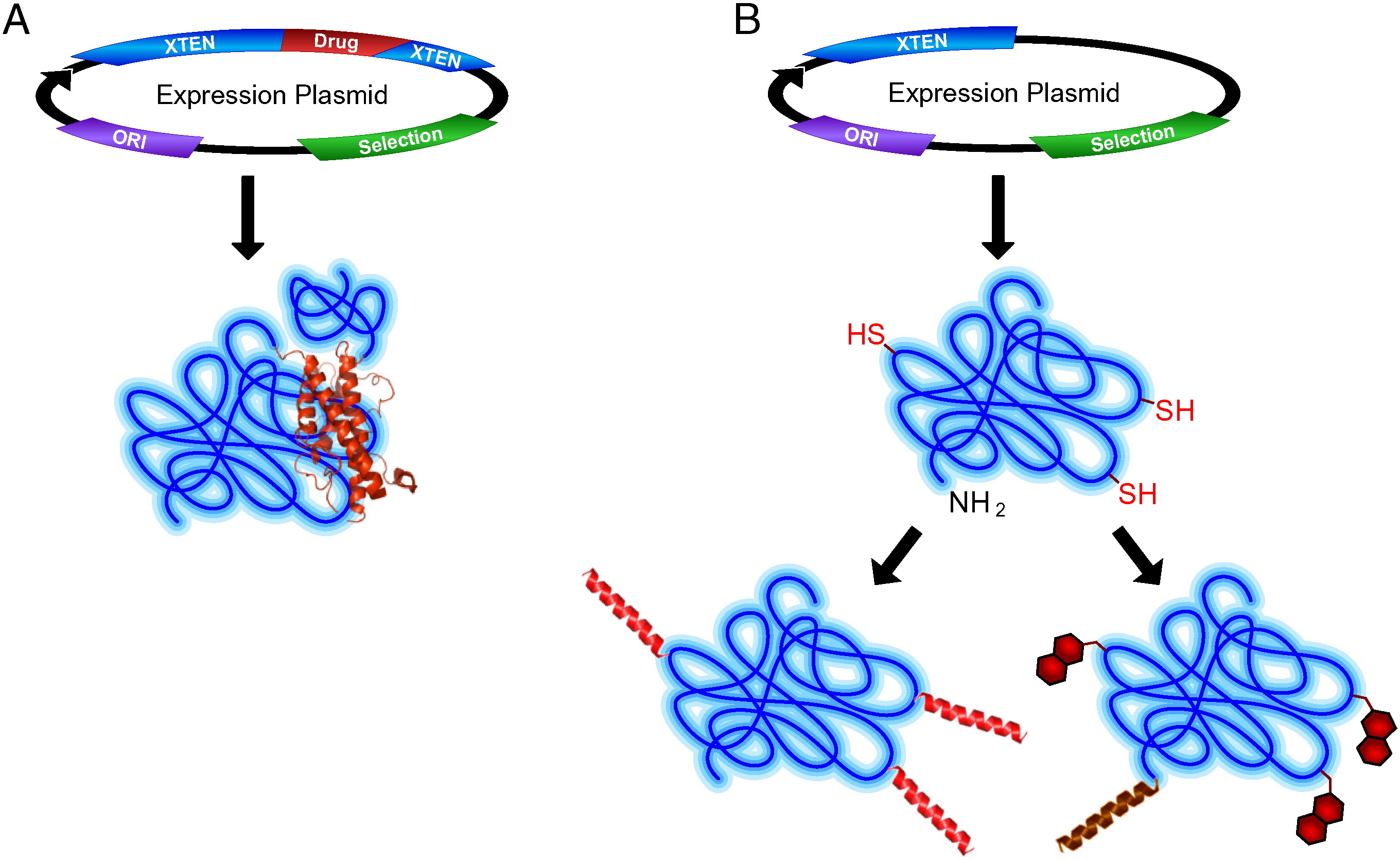 Figure 1. XTENylation of biologically active molecules. Panel A: Genetic fusion. Panel B: Chemical conjugation. (Podust, 2016)
Figure 1. XTENylation of biologically active molecules. Panel A: Genetic fusion. Panel B: Chemical conjugation. (Podust, 2016)
Due to these advantages, XTENylation has been successfully applied in the development of long-acting biologics, including hormones, coagulation factors, and therapeutic peptides, and is increasingly recognized as a powerful tool in biobetter development.
Profacgen provides a comprehensive and flexible XTENylation platform, designed to support the development of optimized long-acting protein and peptide therapeutics. Our services cover the entire workflow from design to validation.
| Steps | Features |
|---|---|
| Strategy Selection and Feasibility Analysis | We begin with a detailed evaluation of the target molecule, considering its structure, function, and therapeutic objectives. Based on this analysis, we recommend the most suitable XTENylation strategy, including fusion orientation, polymer length, and modification approach. |
| XTEN Design and Molecular Engineering |
|
| Genetic Fusion-Based XTENylation | This is the most commonly used approach and involves the direct fusion of XTEN to the therapeutic protein:
|
| Chemical Conjugation-Based XTENylation | For applications requiring additional flexibility, we offer chemical conjugation services:
|
| Expression and Purification |
|
| Functional Characterization and Validation | To ensure the success of XTENylation, we provide extensive analytical and functional evaluation:
|
| PK/PD Evaluation Support | Where required, we support pharmacokinetic and pharmacodynamic studies to confirm extended half-life and improved performance:
|
Depending on your molecule type and therapeutic goals, alternative or complementary strategies may provide additional advantages:
Client Requirements:
A peptide drug candidate exhibited rapid renal clearance and required frequent dosing, limiting its clinical utility and patient compliance. The client needed a half-life extension strategy that would preserve the peptide's receptor-binding activity while significantly improving its pharmacokinetic profile.
Our Solution:
Profacgen designed an XTEN fusion construct with optimized polymer length to increase hydrodynamic size and reduce renal clearance while maintaining the peptide's native conformation. Multiple linker designs were evaluated to ensure proper spatial separation between the peptide and XTEN domains. The fusion protein was expressed in E. coli, purified to high homogeneity, and characterized for binding affinity.
Final Results:
The XTENylated peptide demonstrated significantly prolonged half-life in PK studies, with circulation time extended more than 20-fold compared to the unmodified peptide. Stability against proteolytic degradation was markedly improved, while receptor-binding assays confirmed fully retained biological activity. The client advanced this candidate with confidence in reduced dosing frequency.
Client Requirements:
A therapeutically relevant enzyme showed poor solubility and severe aggregation during bacterial expression, resulting in low yields and inactive protein that hampered downstream development efforts.
Our Solution:
XTEN fusion technology was applied to enhance solubility and stabilize the enzyme during production. The XTEN polypeptide was genetically fused to the enzyme terminus using an optimized linker to minimize structural interference. Expression conditions including temperature and induction timing were optimized to maximize soluble yield.
Final Results:
The XTEN-modified enzyme exhibited dramatically improved solubility with minimal aggregation, enabling high-yield recovery from the soluble fraction. Purified protein maintained full enzymatic activity in functional assays, with stability significantly enhanced during storage. The client successfully utilized this material for biochemical characterization and advanced toward preclinical studies with a robust production platform.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.