Protein co-expression is a powerful strategy for producing multi-protein complexes that are essential for understanding biological systems and developing advanced biotechnological and therapeutic applications. By simultaneously expressing multiple proteins within a single host system, researchers can reconstruct native-like protein assemblies, enabling accurate structural and functional studies.
At Profacgen, we provide a comprehensive protein co-expression platform that integrates advanced vector design, multi-system expression capabilities, and rigorous downstream validation. Our services span bacterial, insect, and mammalian expression systems, offering flexible and scalable solutions for generating functional protein complexes. With extensive experience and optimized workflows, we deliver high-quality, soluble protein assemblies tailored to research, industrial, and preclinical needs.
Multi-protein complexes play indispensable roles in a wide range of cellular processes, including transcription, translation, RNA splicing, signaling cascades, protein trafficking, and degradation. These complexes are responsible for orchestrating biological functions through highly coordinated interactions among multiple protein components.
With the deepening of research in molecular biology and structural biology, it has become increasingly clear that the function of a protein is not solely determined by its intrinsic physicochemical properties. Instead, protein activity often depends on interactions with binding partners, conformational dynamics within complexes, and the overall architecture of multi-component assemblies.
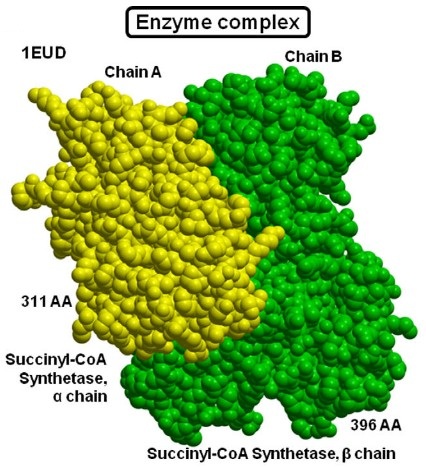 Figure 1. Example of protein–protein complex structure: An enzyme complex (PDB: 1EUD) formed between succinyl-CoA synthetase a chain and succinyl-CoA synthetase b chain is shown in yellow and green colors, respectively. (Sowmya et al., 2015)
Figure 1. Example of protein–protein complex structure: An enzyme complex (PDB: 1EUD) formed between succinyl-CoA synthetase a chain and succinyl-CoA synthetase b chain is shown in yellow and green colors, respectively. (Sowmya et al., 2015)
As a result, there has been a growing emphasis on studying proteins in their native complex forms rather than as isolated molecules. This shift has driven the development of protein co-expression technologies, which enable the simultaneous production of multiple interacting proteins within the same host system.
Protein co-expression offers several critical advantages:
Today, protein co-expression has become a recognized and powerful strategy widely used in:
The ability to reliably co-express multiple proteins in soluble, functional forms is therefore essential for advancing both fundamental research and applied biotechnology.
Profacgen provides a comprehensive, one-stop protein co-expression platform, designed to support the efficient production of multi-protein complexes across diverse expression systems. Our services integrate advanced molecular design, optimized expression strategies, and robust validation to ensure high success rates.
Project Design and Strategy Development
Vector Construction and Cloning
Protein Co-Expression
Protein Purification and Complex Isolation
Quality Control and Functional Validation
Advanced and Optional Services
| Systems | Features | Capabilities | Applications |
|---|---|---|---|
Protein Co-Expression in E. coli |
Co-expression of multi-protein complexes in E. coli provides a cost-effective and efficient alternative to more complex systems such as insect or mammalian cells. |
|
|
Protein Co-Expression in Insect Cells |
Using the baculovirus expression vector system (BEVS), insect cells enable the production of more complex and properly folded protein assemblies with post-translational modifications. |
|
|
Protein Co-Expression in Mammalian Cells |
Mammalian expression systems provide the most physiologically relevant environment for producing complex protein assemblies with native post-translational modifications. |
|
|
Client Requirements:
A research group needed to reconstitute a three-subunit enzyme complex for structural analysis by crystallography. Individual expression of each subunit in E. coli resulted in insoluble inclusion bodies, preventing complex assembly and stalling the client's project timeline.
Our Solution:
We designed a polycistronic vector for simultaneous co-expression of all three subunits in E. coli, ensuring stoichiometric production. Chaperone-assisted folding strategies were incorporated to promote proper conformation, and expression conditions including temperature, IPTG concentration, and media composition were systematically optimized to enhance solubility of the multi-subunit complex.
Final Results:
The optimized approach successfully yielded soluble, correctly assembled enzyme complex with all three subunits present in proper stoichiometry. Purification via affinity and size-exclusion chromatography produced homogeneous material suitable for crystallography trials. The client obtained high-quality complex for structural studies, significantly accelerating their research timeline.
Client Requirements:
The client aimed to study interactions between multiple human signaling proteins that require native post-translational modifications for proper function, which bacterial systems could not provide. They needed correctly modified, assembled complex for interaction and signaling studies.
Our Solution:
We implemented transient co-transfection in HEK293 cells using optimized plasmid ratios to achieve balanced expression of all subunits. Purification strategies were carefully designed to maintain complex integrity throughout the process, incorporating gentle elution conditions and stabilizing buffer components.
Final Results:
The purified protein complex exhibited correct assembly with all subunits present and proper post-translational modifications confirmed by mass spectrometry. Functional activity assays demonstrated the complex retained native signaling capabilities, enabling successful downstream interaction studies and advancing the client's understanding of the signaling pathway.
Background:
A biotech company required a multi-protein viral complex for vaccine research, specifically a properly folded and assembled particle mimicking the native viral structure to support preclinical development and immunogenicity studies.
Our Solution:
We utilized a baculovirus co-infection strategy in insect cells with multiple monocistronic constructs, allowing independent titration of each subunit. Multiplicity of infection (MOI) and harvest timing were systematically optimized to maximize yield while ensuring proper complex assembly and post-translational modifications.
Final Results:
High-yield production of properly folded, functional viral protein complex was achieved, with correct assembly confirmed by electron microscopy and biophysical characterization. The client received milligram quantities of high-quality material that successfully supported immunogenicity studies and advanced their preclinical vaccine development efforts.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.