Membrane proteins—including receptors, transporters, ion channels, and adhesion molecules—play central roles in cellular communication, signal transduction, and molecular transport. Despite their biological and pharmaceutical importance, membrane proteins remain among the most technically challenging biomolecules to isolate and purify in a native, functional form. Profacgen offers comprehensive membrane protein isolation and purification services designed to preserve structural integrity, biological activity, and stability. By integrating optimized solubilization strategies, membrane mimetic systems, and advanced chromatography workflows, we deliver high-quality membrane proteins suitable for functional assays, structural biology, antibody discovery, and drug screening applications.
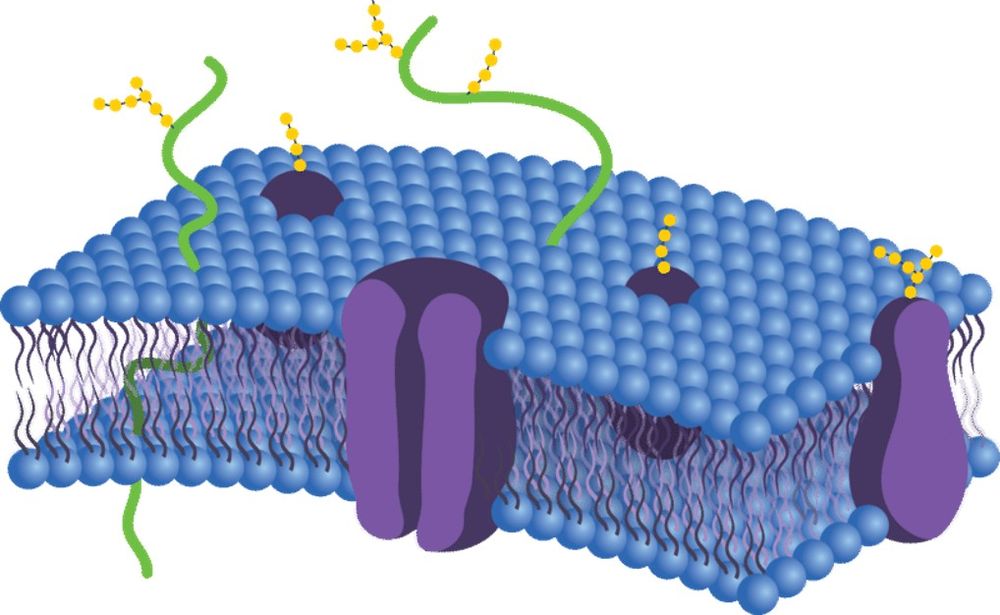
Membrane proteins are embedded within lipid bilayers and contain one or multiple hydrophobic transmembrane domains. Based on secondary structure, they are generally classified into α-helical or β-barrel membrane proteins. These hydrophobic regions are essential for function but introduce major challenges during extraction and purification.
More than 60% of approved drug targets are membrane proteins, underscoring their significance in disease research and therapeutic development. However, unlike soluble proteins, membrane proteins rely on the lipid bilayer to maintain their native conformation. Once removed from the membrane, they are prone to aggregation, misfolding, or irreversible denaturation.
Traditional purification strategies developed for water-soluble proteins are often ineffective for membrane proteins. Successful isolation requires carefully selected detergents or membrane mimetics that can replace the lipid environment while maintaining protein stability. Factors such as detergent chemistry, concentration, temperature, extraction time, and buffer composition all critically influence yield and functionality. Without systematic optimization, membrane protein purification often results in low recovery and compromised activity.
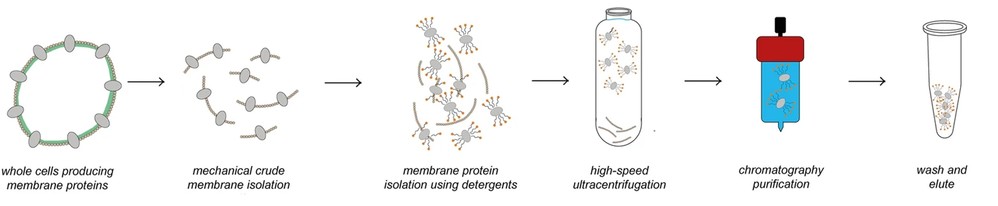 Figure 1. Traditional method for membrane proteins purification. (Adapted from Hering et al., 2020)
Figure 1. Traditional method for membrane proteins purification. (Adapted from Hering et al., 2020)
Profacgen has established a dedicated membrane protein platform supporting detergent-based extraction as well as alternative membrane mimetic systems. With access to more than 100 solubilization agents—including nonionic, zwitterionic, and ionic detergents—we tailor solubilization strategies based on protein topology, size, and biochemical properties.
Available solubilization approaches include:
Customized Experimental Design
Our scientists design each purification workflow based on the specific characteristics of the target membrane protein. Optimization parameters include:
This rational, data-driven approach minimizes protein loss and preserves native structure.
Multi-Modal Purification and Quality Control
Following solubilization, membrane proteins are purified using scalable chromatography techniques, including:
Gel-based and gel-free analytical QC—such as SDS-PAGE, Western blotting, SEC profiling, and activity assays—ensure that purified proteins meet client-defined standards.
Flexible Scale and Target Compatibility
Whether the target is a single-pass receptor, multi-pass GPCR, membrane enzyme, or large protein complex, Profacgen supports:
Our platform provides consistent solutions from exploratory research through advanced development stages.
Background
A pharmaceutical company required purified G protein–coupled receptor (GPCR) protein for ligand-binding and functional screening assays. The target GPCR was unstable during conventional detergent extraction, resulting in aggregation and loss of binding activity, which compromised assay reliability.
Our Solution
Profacgen performed systematic screening of mild non-ionic detergents, cholesterol analogs, and stabilizing additives to identify conditions preserving receptor integrity. Affinity chromatography was combined with size exclusion chromatography (SEC) to enrich monodisperse protein populations while removing aggregates and degraded species.
Final Results
The purified GPCR maintained native ligand-binding activity and showed excellent thermal and temporal stability. The protein was successfully applied in downstream pharmacological assays, enabling reproducible screening and hit identification.
Background
An academic research group aimed to obtain high-purity, structurally intact multi-pass ion channel protein for cryo-electron microscopy (cryo-EM) studies. Previous purification attempts resulted in heterogeneous samples with poor particle quality, limiting achievable resolution.
Our Solution
We developed a mixed-detergent extraction strategy to balance solubilization efficiency and protein stability, followed by controlled reconstitution into nanodiscs to mimic the native lipid environment. Sequential affinity purification and SEC were applied to isolate homogeneous ion channel complexes suitable for structural analysis.
Final Results
The final preparation displayed excellent monodispersity and structural integrity. Cryo-EM analysis yielded well-defined particles and enabled high-resolution structure determination, supporting detailed functional and mechanistic studies of the ion channel.
Background
A biotechnology company required native transporter protein antigens for antibody discovery campaigns. The target protein contained multiple transmembrane helices and conformational epitopes that were easily disrupted during extraction, leading to poor antibody specificity in early screening efforts.
Our Solution
Profacgen optimized detergent composition, extraction temperature, and buffer additives to preserve extracellular domain conformation. A gentle affinity purification workflow was implemented to minimize structural stress, followed by polishing steps to remove misfolded or partially solubilized species.
Final Results
The purified transporter protein retained native conformation and functional epitopes. It was successfully used in immunization and in vitro screening, enabling the isolation of high-affinity, conformation-sensitive antibodies suitable for downstream functional validation.
Reference:
Fill out this form and one of our experts will respond to you within one business day.