Stable cell line development is a cornerstone of modern biologics production, enabling consistent, scalable, and high-yield expression of recombinant proteins, monoclonal antibodies, and other therapeutic molecules. However, traditional workflows are often time-consuming, labor-intensive, and difficult to scale.
Profacgen offers integrated Cell Line Development services that combine advanced screening technologies, optimized expression systems, and deep bioprocess expertise to deliver high-producing, stable, and regulatory-compliant cell lines. Our solutions span early-stage research to GMP-grade development, ensuring seamless transition from discovery to commercial manufacturing.

Stable cell lines are widely used in a number of important applications including biologics (e.g. recombinant protein and monoclonal antibody) production, drug screening, and gene functional studies.
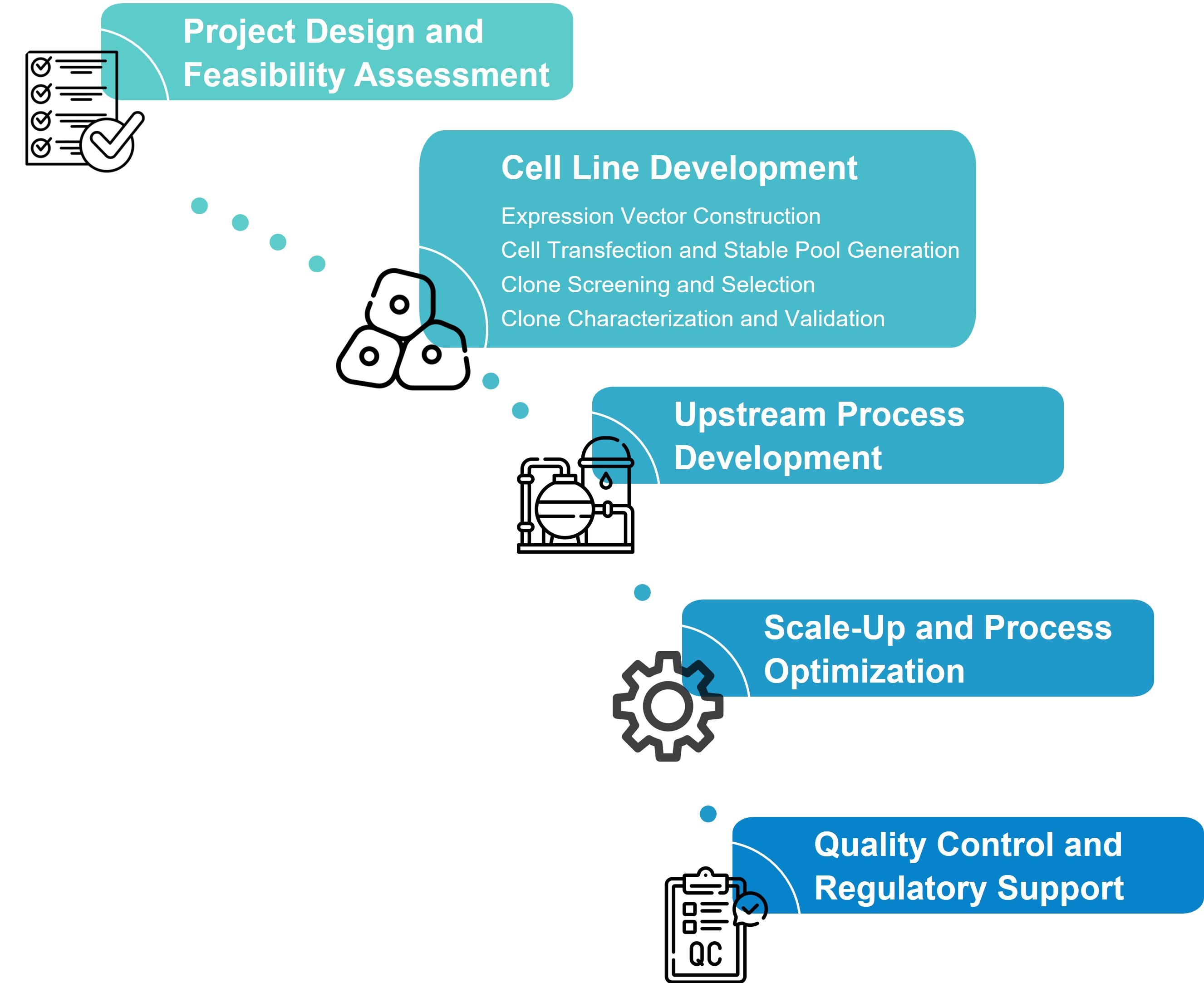
The process of developing stable cell lines typically involves several key stages:
Despite its importance, traditional cell line development relies heavily on manual screening and iterative optimization, making it:
These challenges have driven the need for high-throughput, automated, and data-driven approaches that accelerate development while improving reproducibility and scalability.
Profacgen's development services take advantage of advanced technology and proven expertise to help customers meet their biologics development needs. Our cell line development team collaborates closely with our bioanalytical testing and manufacturing teams to provide high-performance, highly stable, production cell lines in a wide variety of mammalian host cells within the shortest development cycle.
We provide three major service modules:
| Service Module | Overview | Core Capabilities | Best For |
|---|---|---|---|
| Stable Cell Line Construction for Protein Expression | Generation of high-yield, stable expression cell lines for recombinant proteins and antibodies using optimized mammalian systems. |
|
Early-stage research, recombinant protein expression, antibody discovery, difficult-to-express targets |
| Cell Line Development for Industrial Manufacturing | Optimization and selection of production-ready cell lines with strong scalability and process compatibility for large-scale manufacturing. |
|
Process development, scale-up production, preclinical to commercial manufacturing transition |
| GMP-Grade Stable Cell Line Development | Regulatory-compliant cell line development supporting clinical trials and commercial biologics production. |
|
IND-enabling projects, clinical-stage programs, commercial biologics manufacturing |
Challenge:
A client required a stable CHO cell line capable of producing a monoclonal antibody at high titer with consistent glycosylation profiles for preclinical and eventual clinical development.
Approach:
Profacgen optimized vector design incorporating strong promoters and an advanced GS selection system to drive high expression. Following transfection, we performed high-throughput screening of over 1,000 clones to identify top performers. Lead clones were evaluated for stability, productivity, and glycosylation consistency during fed-batch culture.
Outcome:
Achieved >5 g/L expression in fed-batch culture with consistent N-glycan profiles across clones. Stable expression was maintained over 60+ generations, ensuring robust production. The lead clone was successfully scaled and transferred to a 200 L bioreactor, meeting the client's quality requirements for ongoing development.
Challenge:
A client faced low expression levels and protein instability when producing a complex recombinant protein using standard systems. These challenges prevented sufficient material for downstream functional studies and preclinical evaluation.
Approach:
Profacgen evaluated multiple host systems, including CHO and HEK293, to identify the optimal platform. We optimized codon usage, signal peptides, and expression vector design to enhance translation and secretion. A temperature shift strategy was implemented during fed-batch culture to promote proper folding and reduce aggregation.
Outcome:
Achieved an 8-fold increase in expression compared to initial conditions. The optimized process significantly improved protein folding and stability, with reduced aggregation confirmed by analytical SEC. High-quality purified material enabled successful downstream purification and advanced the client's preclinical program.
Consult Our Experts on Your Project
Please do not hesitate to contact us for more details of our cell line development services. Our experts will provide and help design an optimal development solution tailored to your project goals.
Fill out this form and one of our experts will respond to you within one business day.