Co-expression in insect cells using the Baculovirus Expression Vector System (BEVS) is a highly effective strategy for producing complex, multi-protein assemblies with eukaryotic folding and post-translational modifications. By enabling the simultaneous expression of multiple foreign genes within a single host cell, BEVS supports the formation of biologically active protein complexes that closely resemble their native structures.
At Profacgen, we provide advanced co-expression services in insect cells based on single multi-gene baculovirus systems, delivering reproducible, homogeneous, and high-yield expression of protein complexes. Our platform integrates multi-gene vector design, expression optimization, and rigorous quality validation to support applications in structural biology, vaccine development, and biopharmaceutical research. With extensive expertise in baculovirus engineering, we ensure efficient production of functional multi-protein assemblies tailored to your project needs.
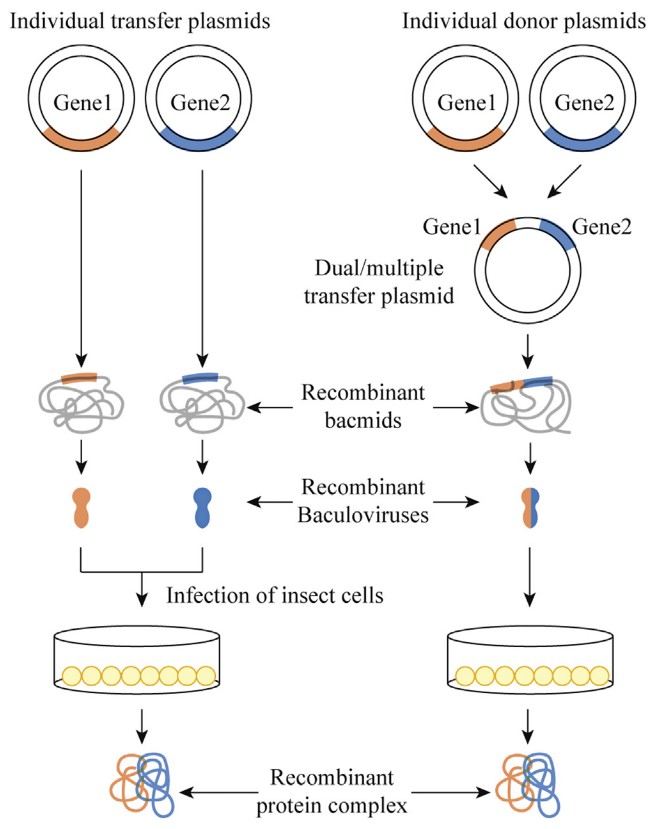 Figure 1. Strategies for expression of multicomponent protein complexes using baculoviruses. (Marintcheva, 2018)
Figure 1. Strategies for expression of multicomponent protein complexes using baculoviruses. (Marintcheva, 2018)
Multi-protein complexes play essential roles in numerous cellular processes, including transcription, translation, signaling cascades, protein trafficking, and degradation. These complexes act as coordinated molecular systems, where multiple protein components interact dynamically to regulate biological functions.
As research has progressed, it has become increasingly clear that proteins do not function in isolation. Instead, their biological activity depends heavily on interactions with partner proteins, the structural organization of complexes, and the cellular environment in which they operate. Consequently, studying proteins in the context of their native complexes has become a central focus in modern life sciences.
Reconstructing these complexes in vitro can be challenging due to issues such as improper folding, instability, or inefficient assembly. Protein co-expression has emerged as a powerful solution, enabling the simultaneous production and in vivo assembly of multiple proteins within a single host system.
The Baculovirus Expression Vector System (BEVS) has become one of the most widely used platforms for co-expression of multi-protein complexes. Insect cells, such as Sf9 and High Five, provide several advantages:
In BEVS, multi-protein expression can be achieved through two main strategies:
While co-infection offers flexibility, it introduces variability in infection efficiency and gene dosage. In contrast, co-expression using multi-gene vectors provides better control over gene expression ratios and ensures homogeneous protein production, making it the preferred strategy for many applications.
Profacgen provides a comprehensive and advanced co-expression service in insect cells, centered on multi-gene baculovirus systems. Our services are designed to deliver consistent, high-quality protein complexes with optimized expression levels and functional integrity.

Profacgen specializes in the development of multi-gene expression vectors, which enable co-expression of multiple proteins from a single baculovirus.
Key Features
Advantages
Our co-expression service in insect cells supports a wide range of applications:
Client Requirements:
A research group required a stable multi-protein complex for cryo-EM analysis, with precise stoichiometry between subunits essential for high-resolution structural determination. Previous attempts yielded heterogeneous assemblies unsuitable for advanced imaging.
Our Solution:
Profacgen designed a multi-gene baculovirus vector incorporating optimized promoters for each subunit to ensure balanced, coordinated expression. Infection conditions including MOI, cell density, and harvest timing were systematically optimized in Sf9 cells to maximize yield of correctly assembled complex while minimizing degradation and aggregation.
Final Results:
Successfully produced a homogeneous and stable protein complex with correct subunit stoichiometry confirmed by mass photometry. Negative-stain EM verified uniform particle morphology, enabling the client to proceed with high-resolution cryo-EM analysis and achieve near-atomic resolution structure determination.
Client Requirements:
A biotech company aimed to produce virus-like particles (VLPs) for vaccine research, requiring correctly assembled structural proteins that authentically mimic native viral architecture to elicit protective immune responses.
Our Solution:
We constructed a multi-gene baculovirus encoding multiple viral structural proteins, with promoter optimization to achieve appropriate expression ratios for proper self-assembly. Expression parameters including infection timing, temperature, and harvest windows were carefully optimized in insect cells to maximize VLP formation.
Final Results:
Achieved efficient assembly of VLPs with correct morphology confirmed by transmission electron microscopy and dynamic light scattering. High-yield production provided sufficient material for immunogenicity studies, where particles demonstrated strong antibody responses supporting the client's vaccine development pipeline.
Client Requirements:
The client faced low expression and severe misfolding of a therapeutically relevant target protein when expressed alone, preventing functional studies and downstream applications.
Our Solution:
Profacgen co-expressed the target protein with a customized set of molecular chaperones using a multi-gene baculovirus system. Chaperone selection and expression timing were optimized to promote proper folding while minimizing cellular stress. Infection conditions including chaperone induction and harvest time were systematically evaluated.
Final Results:
Co-expression with chaperones dramatically improved protein folding and solubility, with correctly folded protein confirmed by CD spectroscopy and activity assays. Yield increased significantly, providing sufficient functional material for biochemical characterization and enabling the client's ongoing drug discovery efforts.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.