Polysialylation is an advanced glycoengineering strategy for enhancing the pharmacokinetic performance of protein and peptide therapeutics. By conjugating polysialic acid (PSA)—a naturally occurring, biodegradable, and highly hydrophilic carbohydrate polymer—to biologically active molecules, this approach significantly increases hydrodynamic size, reduces renal clearance, and improves systemic stability.
As a natural alternative to synthetic polymers such as PEG, polysialylation offers distinct advantages, including excellent biocompatibility, low immunogenicity, and biodegradability, making it particularly attractive for next-generation biologics. PSA-modified therapeutics have demonstrated improved circulation time, reduced immunogenic responses, and enhanced therapeutic efficacy.
At Profacgen, we provide comprehensive polysialylation services, covering strategy design, conjugation development, protein production, and functional validation. Our integrated platform supports the development of optimized long-acting biologics for applications ranging from therapeutic proteins to advanced drug delivery systems.
Protein- and peptide-based therapeutics have become indispensable in modern medicine, offering high specificity and potent biological activity. However, many of these molecules suffer from rapid clearance, short plasma half-life, and limited stability, which restrict their clinical effectiveness and require frequent dosing.
To address these challenges, a variety of half-life extension technologies have been developed, including polymer conjugation and protein fusion strategies. Among these, polysialylation has emerged as a promising and biologically inspired approach, leveraging naturally occurring carbohydrate structures to improve pharmacokinetic properties.
Sialic acids are a family of neuraminic acid derivatives that commonly occur at the terminal positions of glycan chains on glycoproteins and glycolipids. Polysialic acid (PSA) consists of linear homopolymers of sialic acid residues, typically linked via α-2,8 or α-2,9 glycosidic bonds.
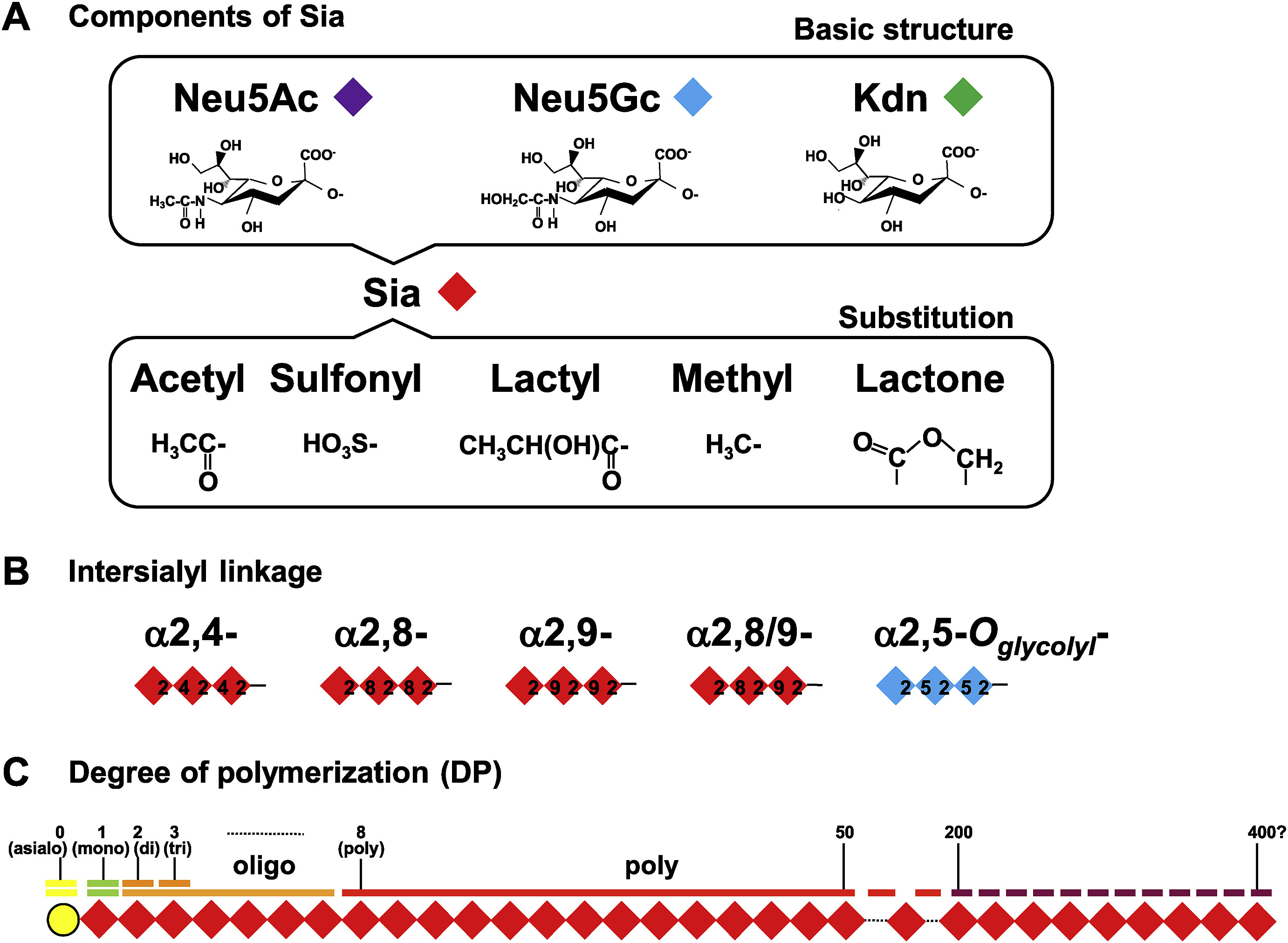 Figure 1. Structures of sialic acid and polysialic acid. A. Structures of sialic acids (Sias). B. Intersialyl linkages of polymerized Sia. C. The degree of polymerization (DP) of polymerized Sia structure. (Sato and Kitajima, 2021)
Figure 1. Structures of sialic acid and polysialic acid. A. Structures of sialic acids (Sias). B. Intersialyl linkages of polymerized Sia. C. The degree of polymerization (DP) of polymerized Sia structure. (Sato and Kitajima, 2021)
PSA is widely found in biological systems, particularly in:
In physiological contexts, PSA plays critical roles in:
Notably, polysialylated NCAM is highly expressed in the embryonic brain, highlighting its importance in developmental processes and disease-related studies.
The unique physicochemical properties of PSA make it highly suitable for drug modification:
Unlike synthetic polymers, PSA is a natural component of glycoproteins, which contributes to its superior biocompatibility and safety profile.
Polysialylation has gained increasing attention as a biologically derived alternative to PEGylation. While PEGylation is widely used, it is associated with potential drawbacks such as non-biodegradability and the emergence of anti-PEG antibodies.
In contrast, PSA offers:
In addition to half-life extension, PSA also demonstrates versatility in drug delivery applications, including use as a sustained-release material, carrier coating, and scaffold for tissue engineering, particularly in nerve repair.
Profacgen provides a comprehensive polysialylation platform, integrating advanced glycoengineering technologies with protein production and analytical capabilities to support the development of optimized therapeutics.

Chemical Conjugation
The most widely used method involves covalent attachment of PSA chains to specific sites on the target protein:
Enzymatic Polysialylation
Where applicable, we offer enzymatic approaches that mimic natural glycosylation processes:
Our polysialylation services support a broad range of applications:
Depending on your molecule type and therapeutic goals, alternative or complementary strategies may provide additional advantages:
Client Requirements:
A peptide drug candidate exhibited rapid renal clearance and required frequent dosing, limiting its clinical utility and patient compliance. The client needed a half-life extension strategy that would improve stability while preserving the peptide's native biological activity.
Our Solution:
Profacgen applied controlled polysialylation using optimized polysialic acid (PSA) chains to increase the peptide's hydrodynamic size and reduce renal clearance. PSA conjugation conditions, including chain length and coupling chemistry, were carefully optimized to maximize half-life extension while minimizing impact on receptor binding. The modified peptide was purified and characterized for PSA content and uniformity.
Final Results:
The polysialylated peptide exhibited significantly prolonged half-life in pharmacokinetic studies, with circulation time extended more than 15-fold compared to the unmodified form. Stability against proteolytic degradation was markedly improved, while receptor-binding assays confirmed fully retained biological activity. The client advanced this candidate with confidence in reduced dosing frequency and enhanced therapeutic potential.
Client Requirements:
A recombinant protein intended for therapeutic use showed severe aggregation and limited solubility during expression and storage, compromising its developability and formulation potential.
Our Solution:
PSA conjugation was implemented to enhance surface hydrophilicity and reduce protein-protein interactions driving aggregation. Polysialic acid chains were site-specifically conjugated to the protein under controlled conditions to maximize solubility improvement while maintaining structural integrity. Conjugation parameters including PSA chain length and molar ratio were systematically optimized.
Final Results:
The polysialylated protein demonstrated dramatically improved solubility with minimal aggregation even at elevated concentrations. Stability studies confirmed enhanced resistance to thermal and storage-induced degradation, while functional assays verified fully maintained biological activity. The client successfully formulated the modified protein for preclinical studies, overcoming previous developability challenges.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.