Insect cell lysate cell-free protein expression is an advanced in vitro protein synthesis platform capable of producing structurally complex and functionally active eukaryotic proteins, including membrane proteins and glycosylated proteins. Based on Spodoptera frugiperda (Sf21) cell extracts, this continuous exchange cell-free system provides a native eukaryotic translation and modification environment. Profacgen's insect cell lysate CFPS platform retains endogenous microsomal vesicles derived from the endoplasmic reticulum, enabling signal peptide processing, membrane insertion, and post-translational modifications. This system is particularly well suited for challenging targets that are difficult to express in conventional cell-based or bacterial cell-free systems.
Cell-free protein expression systems derived from insect cells represent a next-generation CFPS technology designed to overcome limitations of both prokaryotic systems and simpler eukaryotic extracts. Unlike bacterial lysates, insect cell lysates preserve key components of the eukaryotic secretory pathway, including microsomal vesicles originating from the endoplasmic reticulum (ER).
These microsomes provide a functional membrane environment that supports co-translational translocation, signal peptide cleavage, membrane protein insertion, and certain post-translational modifications such as N-linked glycosylation. As a result, insect cell lysate CFPS is especially valuable for expressing membrane proteins, secreted proteins, and multi-domain eukaryotic proteins that require native folding and processing.
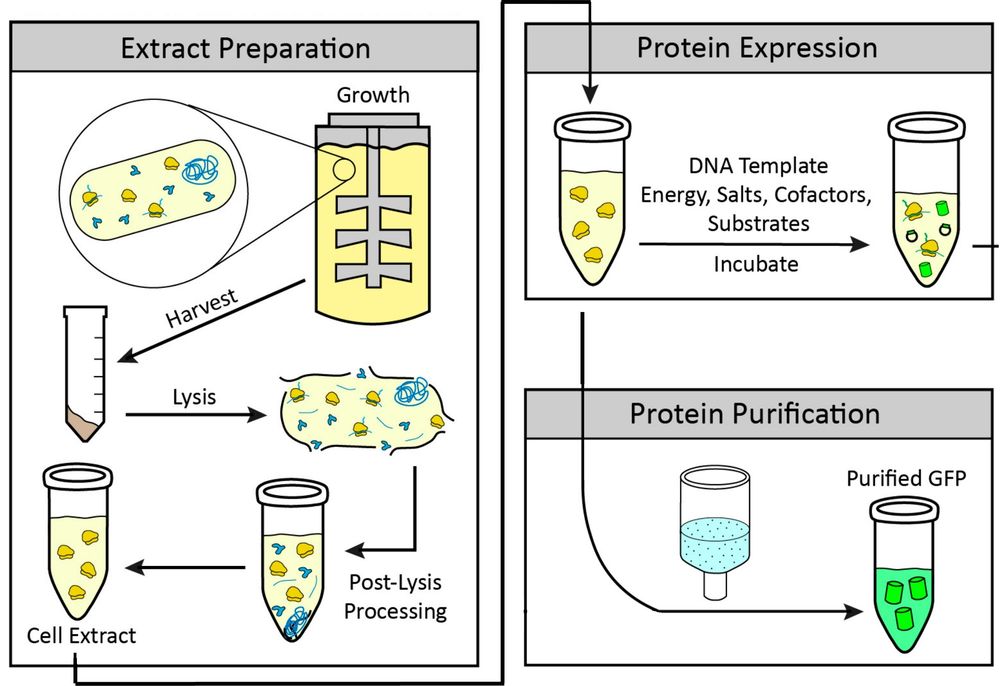 Figure 1. Cell-free protein synthesis. (Adapted from Gregorio et al., 2019)
Figure 1. Cell-free protein synthesis. (Adapted from Gregorio et al., 2019)
Profacgen offers a highly optimized insect cell lysate CFPS platform based on Sf21 extracts, combined with a continuous exchange reaction format and an optional coupled transcription–translation system, enabling efficient, scalable, and reproducible protein production.
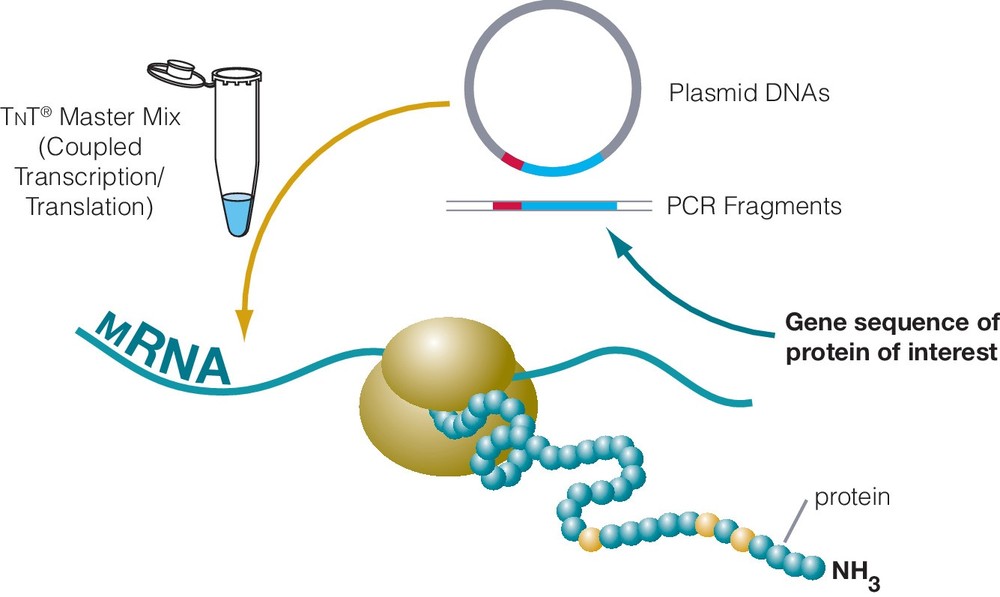 Figure 2. Coupled transcription–translation T7 insect cell extract system.
Figure 2. Coupled transcription–translation T7 insect cell extract system.
Profacgen provides comprehensive insect cell lysate CFPS services tailored to complex protein expression needs:
Expression of Complex and Membrane Proteins
Eukaryotic Post-Translational Modifications
Coupled Transcription–Translation System
Continuous Exchange Cell-Free Expression
High-Throughput and Parallel Protein Production
Protein Purification and Technical Support
| Steps | Details |
|---|---|
| Preparation of Insect Cell Extracts | High-quality Sf21 cell lysates are prepared through gentle cell disruption, preserving translational machinery and endogenous microsomal vesicles. |
| Assembly of the Reaction System | Amino acids, T7 RNA polymerase, energy substrates, and required cofactors are added to the lysate to form the cell-free reaction mixture. Alternatively, Profacgen's coupled transcription–translation system enables a streamlined, single-tube setup. |
| Template Addition and Transcription | Plasmid DNA or PCR-generated templates are added. mRNA is synthesized in vitro under the control of the T7 promoter, with optional companion vectors to enhance translation efficiency. |
| Translation and Protein Synthesis | mRNA is translated into protein using insect-derived ribosomes and translation factors. Signal peptides guide proteins into microsomal vesicles for processing and modification. |
| mRNA Recycling and Reaction Termination | Released mRNA can be reused for multiple rounds of translation, increasing yield. After repeated cycles, mRNA is naturally inactivated, completing the reaction. |
Client Requirement:
A pharmaceutical company required expression of a multi-pass membrane receptor for ligand-binding and signaling studies. Conventional insect cell culture systems produced low yields and inconsistent membrane localization, while bacterial systems failed entirely.
Our Solution:
Profacgen applied the insect cell lysate CFPS platform with preserved ER-derived microsomal vesicles to enable co-translational membrane insertion. A continuous exchange reaction format was used to prolong synthesis and improve yield, while signal peptide design was optimized for efficient translocation.
Final Results:
The receptor was successfully synthesized and correctly inserted into microsomal membranes. Functional assays confirmed ligand binding activity, enabling downstream pharmacological screening without additional optimization.
Client Requirement:
A research institute required production of a secreted, glycosylated eukaryotic protein for antibody development and structural characterization. Expression attempts using E. coli and wheat germ cell-free systems yielded high protein levels but lacked proper signal peptide processing and glycosylation, resulting in reduced immunogenicity and limited suitability for downstream applications.
Our Solution:
Profacgen applied the insect cell lysate cell-free protein expression system, which preserves ER-derived microsomal vesicles to enable co-translational translocation and processing. Signal peptide design and reaction parameters were optimized to promote efficient secretion, protein folding, and N-linked glycosylation while maintaining overall protein stability.
Final Results:
The target protein was obtained in soluble form with correct signal peptide cleavage and consistent glycosylation. The material met quality requirements for immunization, antibody generation, and subsequent analytical validation.
Consult Our Experts on Your Project
→To learn more about the technical approaches and practical applications, visit our detailed guide on Recombinant Protein Expression in Insect Cells Using the Baculovirus Expression System
Reference:
Fill out this form and one of our experts will respond to you within one business day.