Macromolecular protein and peptide therapeutics have become essential tools in modern medicine, with more than 200 recombinant biologics—including peptides, hormones, coagulation factors, enzymes, and growth factors—successfully translated into clinical use. Despite their proven efficacy, a major limitation of many protein therapeutics is their short plasma half-life, often ranging from minutes to less than one day. This rapid clearance necessitates frequent administration, leading to fluctuating drug levels, reduced patient compliance, and increased treatment burden.
Half-life extension technologies provide a powerful solution to these challenges by enhancing the pharmacokinetic (PK) properties of protein therapeutics. Through strategies such as molecular fusion, polymer conjugation, and receptor-mediated recycling enhancement, these technologies can increase circulation time by 2- to 100-fold, significantly improving therapeutic efficacy and dosing convenience.
At Profacgen, we offer a comprehensive suite of half-life extension solutions, combining advanced molecular engineering with rigorous PK/PD evaluation. Our integrated platform enables the design, production, and characterization of optimized long-acting biologics tailored to diverse therapeutic needs.
Protein- and peptide-based therapeutics have revolutionized the treatment of numerous diseases, including metabolic disorders, cancer, autoimmune diseases, and hematological conditions. Their high specificity and biological activity make them attractive alternatives to small-molecule drugs. However, their clinical application is often constrained by unfavorable pharmacokinetic properties.
Many therapeutic proteins are rapidly cleared from circulation due to several physiological mechanisms:
As a result, most protein therapeutics require frequent intravenous (IV) or subcutaneous (SC) administration, often at high doses. This leads to:
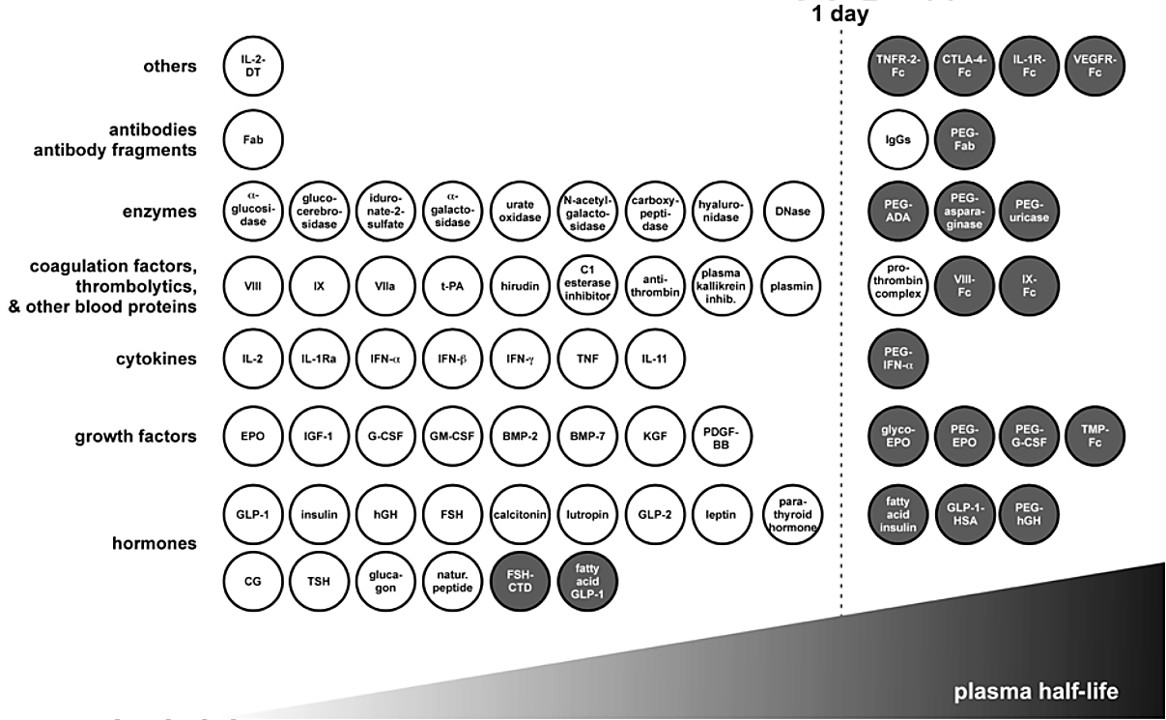 Figure 1. Overview of approved therapeutic proteins and half-life extended derivatives thereof (Kontermann, 2016)
Figure 1. Overview of approved therapeutic proteins and half-life extended derivatives thereof (Kontermann, 2016)
To address these limitations, the pharmaceutical industry has increasingly focused on extending the plasma half-life of biologics. Approximately 15% of approved therapeutic proteins have been engineered with half-life extension strategies, highlighting their importance in modern drug development.
These technologies aim to:
As a result, half-life extension can dramatically improve pharmacokinetic and pharmacodynamic (PK/PD) profiles, enabling less frequent dosing and more consistent therapeutic effects.
Several well-established approaches are widely used in the industry:
These strategies have been successfully applied across a wide range of therapeutic proteins, including hormones, enzymes, cytokines, and antibodies.
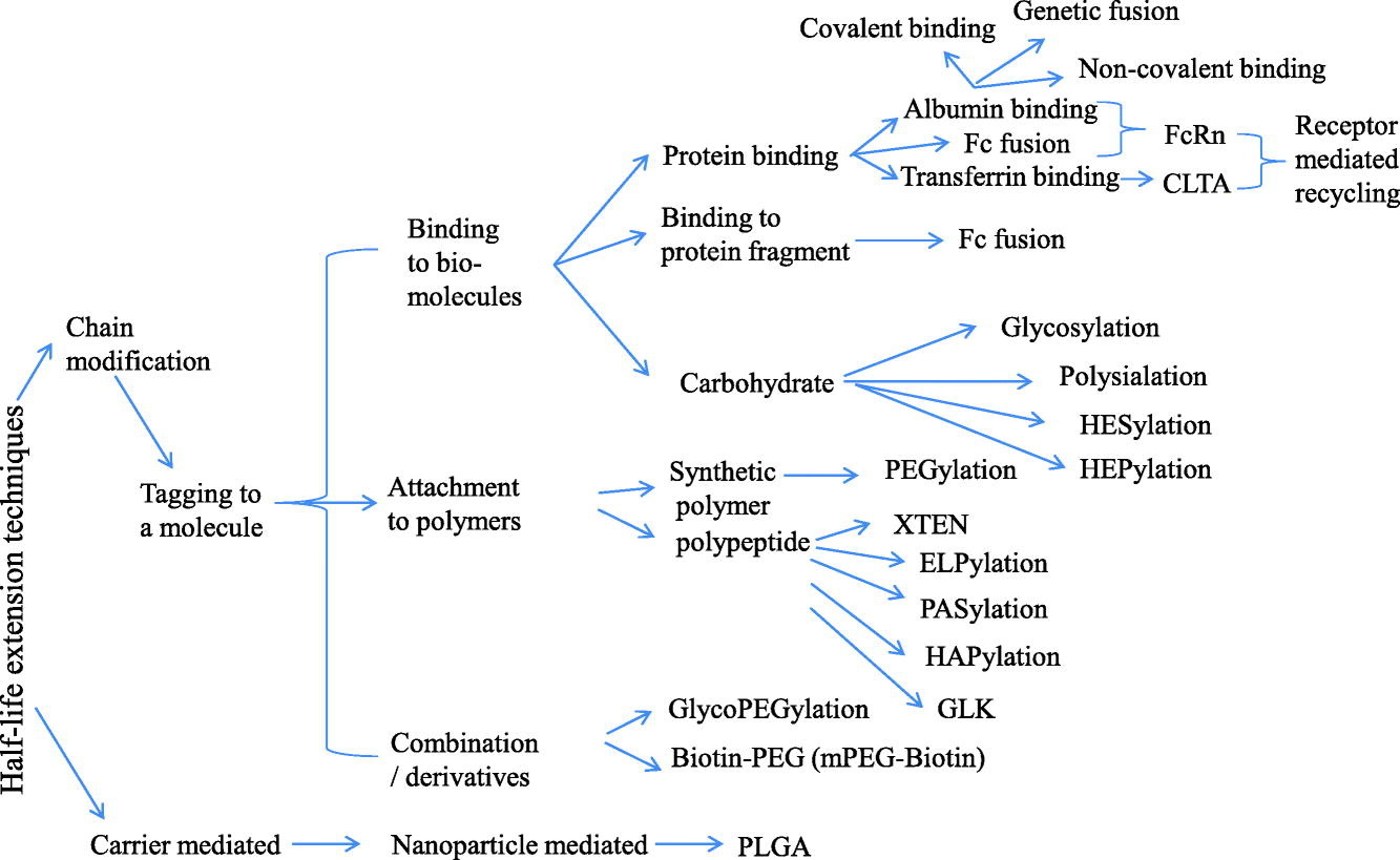 Figure 2. Current strategies in extending half-lives of therapeutic proteins. (Zaman et al., 2019)
Figure 2. Current strategies in extending half-lives of therapeutic proteins. (Zaman et al., 2019)
Profacgen provides a comprehensive, modular platform for half-life extension, covering strategy selection, molecular design, protein production, and in-depth functional evaluation. Our services are tailored to the specific characteristics of each protein and the intended application.

Fc Fusion Protein Production Service
Fusion with the Fc region of immunoglobulins enables interaction with the neonatal Fc receptor (FcRn), which protects proteins from lysosomal degradation and extends circulation time. This approach also improves protein stability and facilitates purification using Protein A/G affinity chromatography.
HSA Fusion Protein Production Service
Human serum albumin (HSA) has an exceptionally long half-life (~19 days) and natural accumulation in certain tissues. Fusion with HSA significantly enhances the half-life and bioavailability of therapeutic proteins while maintaining low immunogenicity.
Serum IgG Fusion Protein Production Service
Fusion to full-length or fragment IgG domains provides both half-life extension and potential effector functions such as antibody-dependent cellular cytotoxicity (ADCC) and complement activation, depending on design.
Attachment of polysialic acid chains increases the hydrodynamic size of proteins, reducing renal clearance and improving stability without significantly affecting biological activity.
Fusion with XTEN, a biodegradable, unstructured polypeptide, increases the hydrodynamic radius of proteins, thereby prolonging circulation time while avoiding some limitations associated with synthetic polymers.
Unnatural Amino Acid Technology for Half-Life Extension
Unnatural amino acid technology enables site-specific protein modification for controlled conjugation, improving stability, bioavailability, and half-life while preserving biological activity and ensuring product homogeneity.
| Feature | Fc Fusion | HSA Fusion | Serum IgG Fusion | Polysialylation | PEGylation | XTENylation | Unnatural Amino Acid |
|---|---|---|---|---|---|---|---|
| Type | Protein fusion (IgG Fc) | Protein fusion (albumin) | Immunoglobulin-based fusion | Natural carbohydrate conjugation | Synthetic polymer conjugation | Genetically encoded polypeptide | Site-specific chemical modification |
| Biodegradability | Biodegradable | Biodegradable | Biodegradable | Fully biodegradable | Non-biodegradable | Fully biodegradable | Depends on conjugate |
| Product Homogeneity | High | High | High | Moderate–High | Often heterogeneous | High | Very high (site-specific) |
| Half-Life Extension Mechanism | FcRn recycling | FcRn recycling | FcRn recycling + immune interaction | Increased hydrodynamic size, reduced clearance | Steric shielding, increased size | Increased size, reduced clearance | Controlled conjugation to half-life extenders |
| Impact on Bioactivity | May alter activity | Usually minimal | May affect function | Minimal (design-dependent) | Can reduce activity | Minimal | Minimal (precise control) |
| Immunogenicity Risk | Low–moderate | Low | Low–moderate | Very low | Moderate | Low | Low |
| Molecular Size Increase | Large (dimeric) | Large | Large | Moderate–large | Variable | Tunable | Minimal–tunable |
| Expression Method | Recombinant | Recombinant | Recombinant | Chemical / enzymatic | Chemical conjugation | Recombinant | Genetic code expansion |
| Manufacturing Complexity | Moderate | Moderate | Moderate–High | Moderate–High | High | Moderate | High |
| Regulatory Familiarity | High | High | Moderate–High | Emerging | High | Emerging | Emerging |
| Best Suited For | Antibody-like biologics | Long-acting proteins | Immune-active biologics | Biocompatible alternatives to PEG | Legacy drugs | Peptides, enzymes | Precision-engineered biologics |
Different technologies are suited for different therapeutic goals:
Client Requirements:
A cytokine therapeutic exhibited rapid renal clearance and required frequent dosing, limiting its clinical utility and patient compliance. The client needed an engineered format to significantly extend half-life while preserving biological activity for ongoing preclinical development.
Our Solution:
Profacgen designed an Fc-fusion construct incorporating the cytokine with an optimized flexible linker to leverage FcRn-mediated recycling and improve stability. The construct was expressed in mammalian cells and purified using Protein A chromatography. Pharmacokinetic (PK) evaluation was conducted to assess half-life extension in relevant models.
Final Results:
The modified cytokine showed a significant increase in half-life and improved in vivo stability compared to the native form. This enabled reduced dosing frequency and enhanced therapeutic potential, supporting the client's advancement toward preclinical development and regulatory studies.
Client Requirements:
A peptide drug for metabolic disease required prolonged circulation to improve efficacy and reduce frequent injections, which were impacting patient adherence and therapeutic outcomes. The client sought a half-life extension strategy that would maintain peptide bioactivity.
Our Solution:
We developed an HSA-fusion construct, optimizing linker design and fusion orientation to preserve the peptide's receptor-binding activity while leveraging albumin's long half-life. The fusion protein was expressed in a mammalian system, purified to high homogeneity, and characterized for binding affinity and stability.
Final Results:
The fusion protein demonstrated markedly improved half-life and bioavailability in PK studies compared to the native peptide, with circulation time extended from hours to several days. Functional assays confirmed fully retained biological activity, supporting further preclinical development and positioning the candidate for advanced efficacy evaluation in disease models.
Client Requirements:
An enzyme therapeutic suffered from rapid proteolytic degradation and poor pharmacokinetic properties, limiting its therapeutic potential and requiring frequent dosing. The client needed a technology to enhance stability without compromising enzymatic activity.
Our Solution:
Profacgen applied XTENylation technology, fusing the enzyme to a long, unstructured hydrophilic polypeptide to increase hydrodynamic size and reduce renal clearance. Multiple XTEN lengths were evaluated to optimize the balance between half-life extension and activity retention. Expression and purification were optimized, followed by functional assays and PK studies.
Final Results:
The optimized XTEN-modified enzyme exhibited significantly prolonged circulation time with half-life extended more than tenfold while maintaining full enzymatic activity. Enhanced stability against proteolytic degradation was confirmed in serum stability assays. This improved therapeutic profile enabled the client to advance the candidate toward preclinical studies with confidence in its dosing regimen and efficacy potential.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.