Precise control of glycosylation has become a defining factor in the development of next-generation biotherapeutics. Among the most impactful glycosylation modifications is core fucosylation, which directly influences the efficacy of therapeutic antibodies. FUT8 knockout (Fut8-KO) CHO-K1 cell lines represent a powerful glycoengineering platform for producing afucosylated or low-fucose proteins and antibodies with enhanced biological activity. By eliminating α-1,6-fucosyltransferase (Fut8) activity, this system enables consistent production of antibodies with significantly improved antibody-dependent cellular cytotoxicity (ADCC). Profacgen provides an end-to-end FUT8-KO CHO-K1 protein and antibody expression service, supporting research, preclinical development, and early manufacturing with high-quality, reproducible, and functionally optimized products.
Fut8 (α-1,6-fucosyltransferase) is the enzyme responsible for catalyzing the addition of fucose to the innermost N-acetylglucosamine (GlcNAc) residue of N-linked glycans, a modification known as core fucosylation. This process is ubiquitous in mammalian expression systems and plays important roles in protein structure, stability, and cellular recognition.
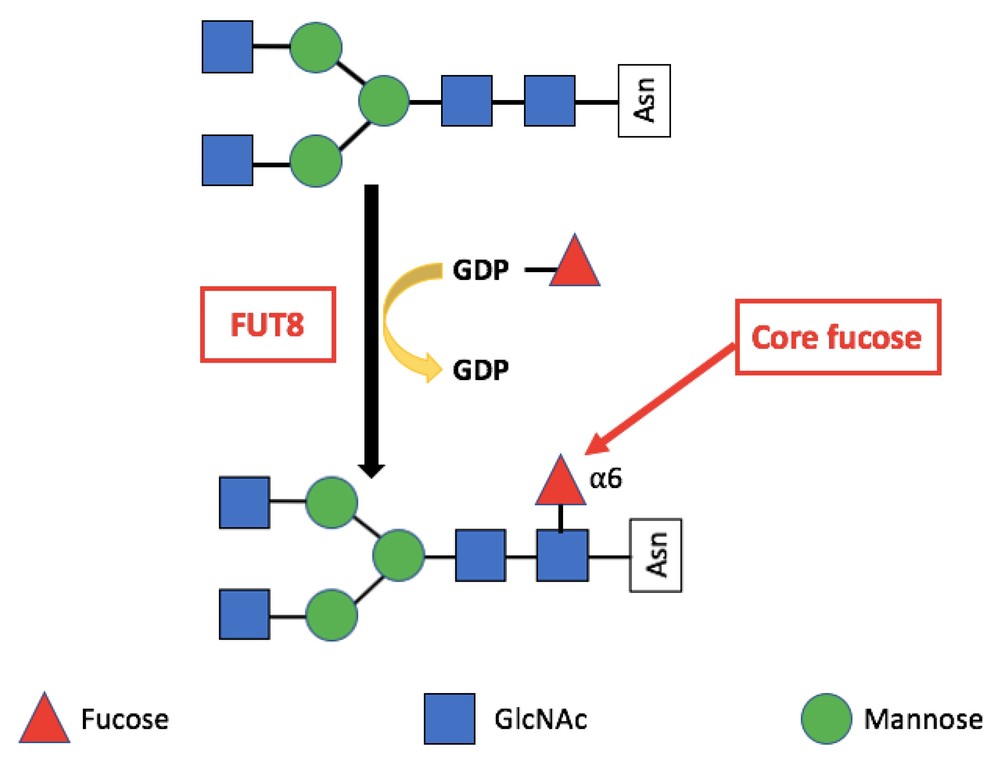 Figure 1. The reaction catalyzed by FUT8. FUT8 transfers an l-fucose reside from GDP-β-l-fucose (GDP-Fuc) onto the innermost GlcNAc of an N-glycan to form an α-1,6 linkage. (Bastian et al., 2021)
Figure 1. The reaction catalyzed by FUT8. FUT8 transfers an l-fucose reside from GDP-β-l-fucose (GDP-Fuc) onto the innermost GlcNAc of an N-glycan to form an α-1,6 linkage. (Bastian et al., 2021)
However, for therapeutic antibodies, particularly IgG1-based biologics, core fucosylation has a critical downside: it reduces binding affinity between the Fc region of the antibody and FcγRIIIa receptors on immune effector cells such as natural killer (NK) cells.
ADCC is a key mechanism of action for many anticancer and antiviral antibodies. Reduced core fucosylation dramatically enhances FcγRIIIa binding, resulting in:
Clinical data have shown that afucosylated antibodies can exhibit up to 10–100-fold higher ADCC activity compared to their fucosylated counterparts.
Chinese Hamster Ovary (CHO) cells remain the gold standard for therapeutic protein production due to their robust growth, human-compatible glycosylation, and regulatory acceptance. By knocking out the FUT8 gene in CHO-K1 cells, it becomes possible to:
 Figure 2. The change of fucosylated glycoproteins in the FUT8KO CHO cells. (A) Heatmap of fucosylated and core-fucosylated glycoproteins in the WT and FUT8KO CHO cells. (B) Percentage of core-fucosylated glycoproteins in fucosylated glycoproteins in the WT and FUT8KO CHO cells. (Yang et al., 2021)
Figure 2. The change of fucosylated glycoproteins in the FUT8KO CHO cells. (A) Heatmap of fucosylated and core-fucosylated glycoproteins in the WT and FUT8KO CHO cells. (B) Percentage of core-fucosylated glycoproteins in fucosylated glycoproteins in the WT and FUT8KO CHO cells. (Yang et al., 2021)
As a result, FUT8-KO CHO-K1 cells have become a cornerstone platform for next-generation antibodies, bispecifics, and Fc-fusion proteins.
Profacgen provides a comprehensive, integrated FUT8-KO CHO-K1 expression platform, covering every stage from cell line development to analytical characterization.
| Services | Details |
|---|---|
| FUT8-KO CHO-K1 Cell Line Development | Gene Editing: FUT8 gene knockout using advanced gene-editing technologies to generate stable, well-characterized FUT8-deficient CHO-K1 cell lines. |
| Cell Line Validation: Confirmation of FUT8 knockout at genomic and functional levels to ensure minimal core fucosylation. |
|
| Stable Cell Line Generation: Long-term genetic stability testing to support reproducible protein production. |
|
| Expression Vector Design and Optimization | Custom vector construction for antibodies, bispecific antibodies, Fc-fusion proteins, and glycoproteins |
| Incorporation of strong promoters, secretion signals, and selection systems (e.g., GS-based selection) | |
| Optimization of heavy and light chain expression balance for antibodies | |
| Transfection, Clone Screening, and Cell Line Selection | Transfection using optimized protocols for FUT8-KO CHO-K1 cells |
| High-throughput clone screening via limiting dilution or FACS | |
| Selection of high-producing clones with stable expression and consistent glycan profiles | |
| Protein and Antibody Expression Optimization | Culture Modes: Fed-batch and perfusion culture strategies optimized for yield and glycan consistency |
| Process Parameters: Optimization of media composition, feeding strategy, temperature, pH, and dissolved oxygen |
|
| Glycoengineering Control: FUT8 knockout ensures reduced fucosylation while maintaining other glycan features such as galactosylation and sialylation |
|
| Analytical Characterization and Quality Control | Glycan Profiling: LC-MS, HILIC-UPLC, MALDI-TOF, and other advanced techniques to confirm afucosylation levels |
| Functional Assays: ADCC, CDC, Fc receptor binding, and potency assays |
|
| Product Quality Testing: Assessment of purity, aggregation, stability, and batch consistency |
|
| Scale-Up and cGMP Readiness | Transition from research-scale to manufacturing-relevant processes |
| Process documentation and data packages suitable for preclinical and early clinical programs | |
| Support for cGMP-compatible workflows |
Background
A biotech company developing an IgG1 monoclonal antibody for oncology required enhanced ADCC activity to improve tumor cell killing. Conventional CHO expression resulted in high fucosylation and suboptimal potency.
Our Solution
We expressed the antibody using a FUT8 knockout CHO-K1 cell line. Expression vectors were optimized for balanced heavy- and light-chain expression, and fed-batch culture conditions were refined to ensure high yield and consistent glycosylation. Glycan profiling and ADCC assays were conducted to verify functional improvement.
Final Results
The antibody showed over 90% reduction in core fucosylation and more than a 20-fold increase in ADCC activity, enabling successful advancement into preclinical development.
Background
A pharmaceutical client was developing a bispecific antibody but encountered batch-to-batch variability caused by heterogeneous glycosylation. Inconsistent fucosylation levels affected Fc receptor binding and led to fluctuating functional readouts, complicating candidate selection and downstream development. The client required a stable expression platform capable of delivering reproducible glycan profiles and consistent biological activity.
Our Solution
We generated a stable FUT8-KO CHO-K1 cell line expressing both antibody arms with optimized gene ratios. Clone screening focused on productivity and glycosylation consistency. Culture parameters were systematically optimized to maintain uniform expression and minimize glycan heterogeneity across batches.
Final Results
The bispecific antibody exhibited highly consistent low-fucose glycosylation, improved Fc receptor engagement, and reproducible functional performance, providing a reliable foundation for further development and scale-up.
Background
A biosimilar developer aimed to produce an antibody comparable to a reference product while enhancing immune effector function to improve clinical differentiation. Conventional CHO expression met basic similarity requirements but offered limited ADCC enhancement. The client needed controlled glycosylation without compromising comparability or regulatory alignment.
Our Solution
The antibody was expressed in FUT8-KO CHO-K1 cells under optimized fed-batch conditions. Comparative glycan profiling, Fc receptor binding studies, and ADCC assays were performed alongside reference product analyses to ensure controlled afucosylation and functional consistency.
Final Results
The final product achieved biosimilarity in critical quality attributes while demonstrating enhanced ADCC activity, supporting both regulatory confidence and improved competitive positioning.
FUT8-KO CHO-K1 expression systems represent a transformative platform for glycoengineered biotherapeutics. By enabling precise control of core fucosylation, this technology unlocks enhanced efficacy, consistency, and flexibility for next-generation protein and antibody development. Profacgen's FUT8-KO CHO-K1 service provides a reliable, scalable, and scientifically rigorous solution to accelerate your biologics program from concept to clinic. Contact us to discuss your project needs.
References:
Fill out this form and one of our experts will respond to you within one business day.