Characterizing the stability of biopharmaceutical proteins is a critical component throughout the entire development lifecycle, including candidate selection, preformulation and formulation development, process optimization, and manufacturing support. Stability data also play an essential role in biocomparability assessments, shelf-life justification, and regulatory decision-making.
Profacgen provides comprehensive protein stability analysis services designed to generate actionable, decision-relevant data that support both early development strategies and regulated programs. Our stability studies are structured to align with downstream analytical, manufacturing, and regulatory requirements, ensuring that generated data retain value across development stages.
Protein stability is governed by a balance of multiple molecular interactions and thermodynamic forces. Among these, three factors are widely recognized as dominant contributors:
Extensive experimental and theoretical studies indicate that hydrophobic interactions and hydrogen bonds are the most prevalent stabilizing forces in folded proteins. During protein folding, hydrophobic moieties become buried within the protein core, while intramolecular hydrogen bonds are formed. This process is accompanied by the release of structured water molecules, contributing favorably to the free energy of folding.
These fundamental interactions underpin higher-order structure, resistance to unfolding, and susceptibility to degradation pathways such as aggregation or chemical modification.
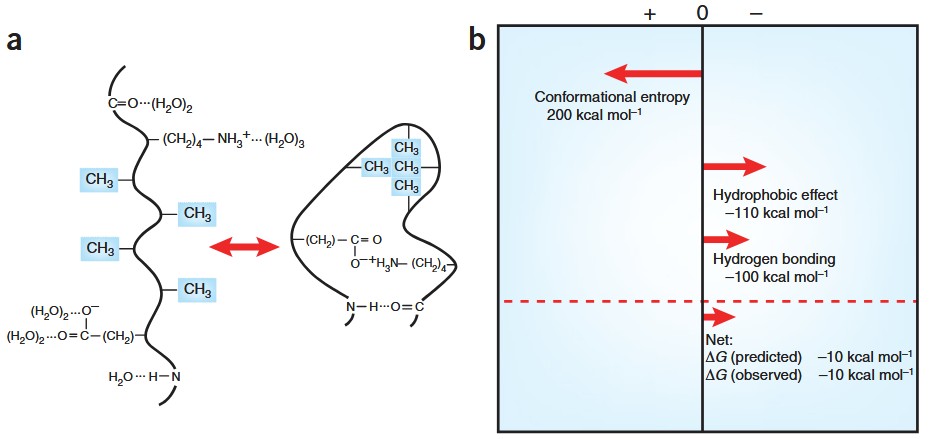 Figure 1. Stabilizing and destabilizing forces on the folding of a protein.(a) Schematic illustrating the formation of an intramolecular hydrogen bond, a hydrophobic bond and an ion pair in the folding of a protein. (b) Rough estimates of the contributions to the free energy of folding of a 100-residue protein. (Pace, 2009)
Figure 1. Stabilizing and destabilizing forces on the folding of a protein.(a) Schematic illustrating the formation of an intramolecular hydrogen bond, a hydrophobic bond and an ion pair in the folding of a protein. (b) Rough estimates of the contributions to the free energy of folding of a 100-residue protein. (Pace, 2009)
While these principles are well established, their practical implications vary significantly between proteins, formulations, and storage conditions—necessitating experimental stability evaluation rather than reliance on theoretical predictions alone.
Protein stability analysis supports multiple critical development decisions, including:
By systematically evaluating stability under defined conditions, clients can anticipate risks, reduce downstream failures, and support regulatory confidence.
Our protein stability analysis services include, but are not limited to:
Each assessment is selected based on its relevance to program objectives rather than applied as a fixed panel.
Profacgen conducts a range of stability studies tailored to program stage and intended application, including:
Study design is guided by scientific rationale, regulatory expectations, and practical development needs.
Stability data are most valuable when interpreted in context. Profacgen integrates stability findings with:
Results are summarized in a format suitable for internal decision-making, cross-functional communication, and regulatory discussion, rather than presented as isolated datasets.
Our goal is not only to measure stability, but to help clients understand and manage stability risk throughout development.
Program Context:
A biopharmaceutical development team was evaluating multiple protein candidates with comparable biological activity but limited stability data. Early identification of stability risks was required to support informed lead selection.
Objective:
To characterize relative stability profiles and identify candidates with acceptable robustness for downstream formulation and manufacturing development.
Approach:
Profacgen designed comparative stability studies, including pH and thermal stress assessments combined with aggregation analysis. Orthogonal analytical techniques were applied to detect early signs of physical or conformational instability under development-relevant conditions.
Outcome:
The resulting data enabled clear differentiation between candidates, supporting selection of a lead molecule with a more favorable stability profile and reduced downstream development risk.
Program Context:
A formulation development program required stability data to evaluate buffer systems and excipient choices for a therapeutic protein intended for refrigerated storage.
Objective:
To assess how formulation variables influenced protein stability and aggregation propensity under both accelerated and real-time conditions.
Approach:
Stability studies were conducted across defined pH ranges and temperature conditions, with aggregation and degradation monitored using stability-indicating analytical methods. Data were integrated to compare formulation performance and identify stabilizing conditions.
Outcome:
The study supported selection of formulation conditions that improved stability and reduced aggregation risk, providing a data-driven basis for further development and scale-up.
Reference:
Fill out this form and one of our experts will respond to you within one business day.