The Tobacco Cell-Free Protein Expression platform is a rapid, animal-free recombinant protein production system designed for targets that are difficult, inefficient, or impractical to express in living cells. By utilizing transcription and translation machinery extracted from tobacco cells, this system enables high-yield protein synthesis without the constraints of cell growth, viability, or cytotoxicity. Proteins such as antibodies, antigens, enzymes, and structurally complex membrane proteins can be produced within hours rather than weeks.
Compared with E. coli-based cell-free system-or wheat germ–based cell-free system, tobacco-derived lysates offer higher expression potential, plant-specific post-translational modification capabilities, and superior biosafety. Profacgen provides a fully integrated service—from gene design and vector construction to expression optimization, validation, and purification—delivering high-quality proteins suitable for functional studies, formulation development, and in vivo applications.
Conventional recombinant protein production relies on living host cells such as bacteria, yeast, insect cells, or mammalian cells. While these systems are effective for many proteins, they frequently encounter limitations when expressing toxic proteins, membrane-associated proteins, or targets with complex folding requirements. Cell viability constraints, long optimization cycles, and low yields often slow research progress and increase costs.
Cell-free protein expression systems (CFS) overcome these limitations by performing transcription and translation entirely in vitro. Essential components—including ribosomes, tRNAs, amino acids, transcription factors, and metabolic enzymes—are preserved in cell lysates, allowing protein synthesis to proceed immediately upon addition of DNA or mRNA templates. Because the system is open and controllable, researchers can directly manipulate reaction conditions, add folding aids or detergents, and fine-tune expression parameters in real time.
Cell-free expression is especially advantageous for rapid prototyping, functional screening, and production of proteins that are unstable or toxic in vivo. For research programs that prioritize speed, flexibility, and experimental control, cell-free systems provide a powerful alternative to traditional cell-based expression platforms.
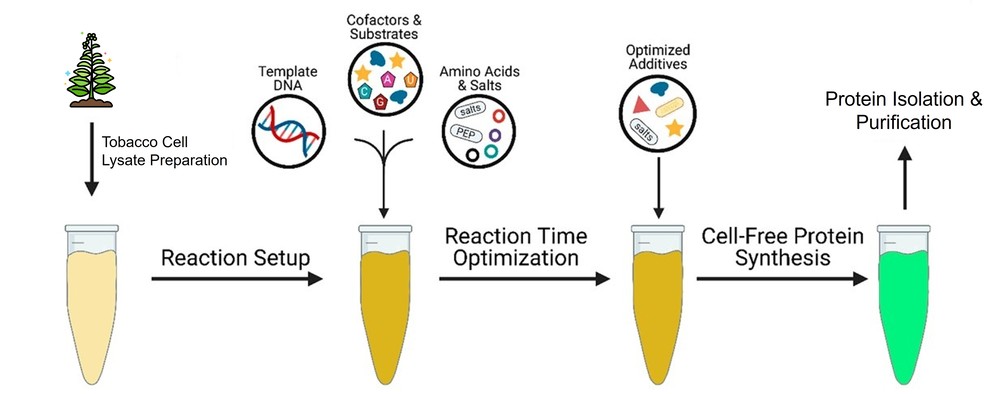 Figure 1. Generalized workflow for cell-free protein synthesis (CFPS). (Adapted from Burrington et al., 2021)
Figure 1. Generalized workflow for cell-free protein synthesis (CFPS). (Adapted from Burrington et al., 2021)
Among available cell-free platforms, tobacco-based systems offer a unique combination of performance, safety, and versatility. Unlike prokaryotic cell-free systems, tobacco lysates originate from a eukaryotic host and therefore retain elements that support more complex protein folding and post-translational processing. At the same time, they remain entirely animal-free, eliminating concerns related to endotoxins, adventitious agents, or animal-derived contaminants.
Compared with wheat germ cell-free systems, tobacco cell-free expression often achieves higher protein yields and supports a broader range of protein classes. The system is particularly well suited for membrane proteins, low-expression targets, and proteins requiring precise experimental control. These advantages make tobacco cell-free expression an attractive option for early-stage discovery, protein engineering, and preclinical research.
The tobacco cell-free expression system is based on a highly active Bright Yellow-2 (BY-2) tobacco cell lysate designed for rapid and reproducible in vitro translation. BY-2 cells are cultivated under controlled suspension culture conditions to ensure consistency and scalability. Cell walls are enzymatically removed to generate protoplasts, followed by a critical evacuolation step that removes lytic vacuoles rich in proteases and nucleases, thereby preserving translational activity.
The purified protoplasts are gently lysed using nitrogen decompression to protect sensitive translation machinery. Endogenous mRNA is eliminated while ribosomal integrity is maintained, resulting in a robust lysate that efficiently translates added templates. The entire BYL preparation can be completed within a few hours, providing a fast, high-performance plant-based cell-free system with superior yield and reproducibility.
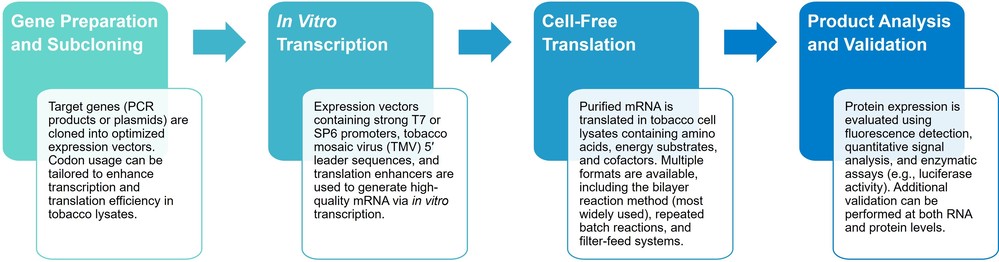
 Figure 2. Flowchart showing the preparation of BYL. (Buntru et al., 2014)
Figure 2. Flowchart showing the preparation of BYL. (Buntru et al., 2014)
Profacgen provides a comprehensive tobacco cell-free protein expression service, including:
Throughout the service process, our scientists work closely with clients to ensure that expression strategies align with experimental objectives and downstream applications.
Client Requirement:
A pharmaceutical research group needed rapid access to a multi-pass membrane protein for ligand-binding and functional screening. Conventional cell-based expression resulted in severe cytotoxicity and protein aggregation, preventing sufficient material production.
Our Solution:
Profacgen applied a tobacco cell-free expression strategy using codon-optimized constructs and a bilayer translation system supplemented with solubilizing additives. Reaction conditions were fine-tuned to balance expression level and protein stability.
Final Results:
Within 24 hours, the target membrane protein was successfully expressed with improved solubility and structural integrity. Purified protein met functional assay requirements, enabling the client to proceed with screening studies without extended host optimization.
Client Requirement:
A biotechnology company developing a recombinant vaccine antigen required an animal-free production platform compatible with downstream in vivo studies. The client needed rapid turnaround, consistent batch-to-batch quality, and minimal regulatory complexity, as the antigen was intended for early immunogenicity evaluation and formulation screening.
Our Solution:
Profacgen expressed the vaccine antigen using the tobacco cell-free protein expression system. Transcriptional elements were optimized to enhance mRNA stability and translation efficiency, while affinity tags and streamlined purification workflows were applied to achieve high purity and reproducibility across reactions.
Final Results:
The recombinant antigen was obtained at high purity, free of endotoxins and animal-derived contaminants. The protein was directly applied in immunization studies, demonstrating reliable performance and significantly reducing preclinical development timelines while simplifying biosafety and regulatory compliance.
Consult Our Experts on Your Project
Profacgen's tobacco cell-free protein expression platform offers a fast, flexible, and animal-free solution for challenging recombinant protein targets. Our experienced scientists are ready to evaluate your project and design an optimized expression strategy tailored to your needs. Contact Profacgen today to explore how tobacco cell-free expression can accelerate your research and development efforts.
References:
Fill out this form and one of our experts will respond to you within one business day.