Detergent selection is a critical determinant of success in membrane protein purification. While detergents enable the solubilization of membrane proteins into aqueous environments, inappropriate choices can destabilize protein structure, disrupt functional conformations, or lead to aggregation and loss of activity. Profacgen provides a systematic detergent screening service designed to identify optimal solubilization conditions for challenging membrane proteins. By combining an extensive detergent library with condition optimization and analytical evaluation, we maximize extraction efficiency while preserving native structure and biological function, supporting downstream applications such as functional assays, structural studies, and antibody discovery.
Detergents, also known as surfactants, are amphiphilic molecules that shield the hydrophobic regions of membrane proteins and allow them to remain soluble in aqueous buffers. Based on the nature of their polar head groups, detergents are commonly classified as ionic, non-ionic, or zwitterionic. Each class exhibits distinct solubilization strength, protein compatibility, and effects on structural integrity.
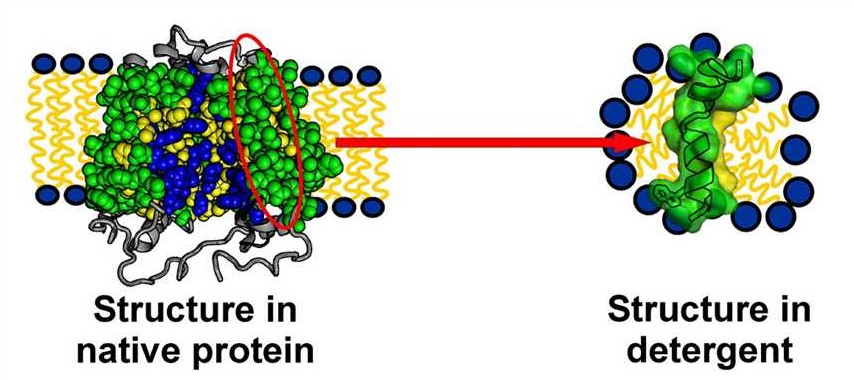
Although detergents are indispensable for membrane protein extraction, they can also interfere with protein stability, folding, and function. Factors such as detergent concentration, micelle size, and critical micelle concentration (CMC) are influenced by experimental conditions including temperature, ionic strength, and buffer composition. Importantly, detergents that efficiently extract a protein are not always suitable for maintaining its native conformation.
There is no universal detergent that works for all membrane proteins. Even closely related targets may respond very differently to the same detergent. As a result, empirical detergent screening remains an essential step in membrane protein purification workflows.
Selecting an appropriate detergent family is a critical early decision in membrane protein purification, as different detergents vary significantly in solubilization strength, micelle size, and impact on protein stability and function. Below we outline the most commonly used detergent categories and their typical use scenarios.
| Category | Description | Best suited for | Advantages | Limitations |
|---|---|---|---|---|
| Maltoside-Based Detergents | Maltosides, such as n-Dodecyl-β-D-maltoside (DDM) and n-Decyl-β-D-maltopyranoside (DM), are among the most widely used detergents for membrane protein research. They are non-ionic, relatively mild, and known for preserving native protein structure and activity. | GPCRs, transporters, ion channels, and proteins requiring long-term stability | High protein compatibility, good functional retention | Larger micelle size may be less optimal for high-resolution structural studies |
| Glucoside-Based Detergents | Glucosides, such as Octyl-β-D-glucoside (OG) and Nonyl glucoside (NG), have shorter alkyl chains and form smaller micelles compared to maltosides. These properties can improve sample homogeneity and reduce background interference. | Crystallography, cryo-EM, and proteins tolerant to stronger detergents | Smaller micelles, easier detergent exchange | Reduced stabilizing capacity for fragile membrane proteins |
| Amine Oxide Detergents | Lauryldimethylamine-N-oxide (LDAO) is a zwitterionic detergent with relatively strong solubilizing power. It is effective for extracting tightly embedded membrane proteins but may compromise stability if not carefully controlled. | Initial extraction of difficult-to-solubilize proteins | High extraction efficiency | Narrow stability window; may disrupt functional conformations |
| Neopentyl Glycol and Polyoxyethylene-Based Detergents | Advanced maltose-neopentyl glycol detergents combine strong stabilization with favorable micelle properties. These detergents have gained popularity in structural biology applications. | Cryo-EM, single-particle analysis, and sensitive multi-pass proteins | Exceptional stability, reduced aggregation | Higher cost and slower detergent exchange |
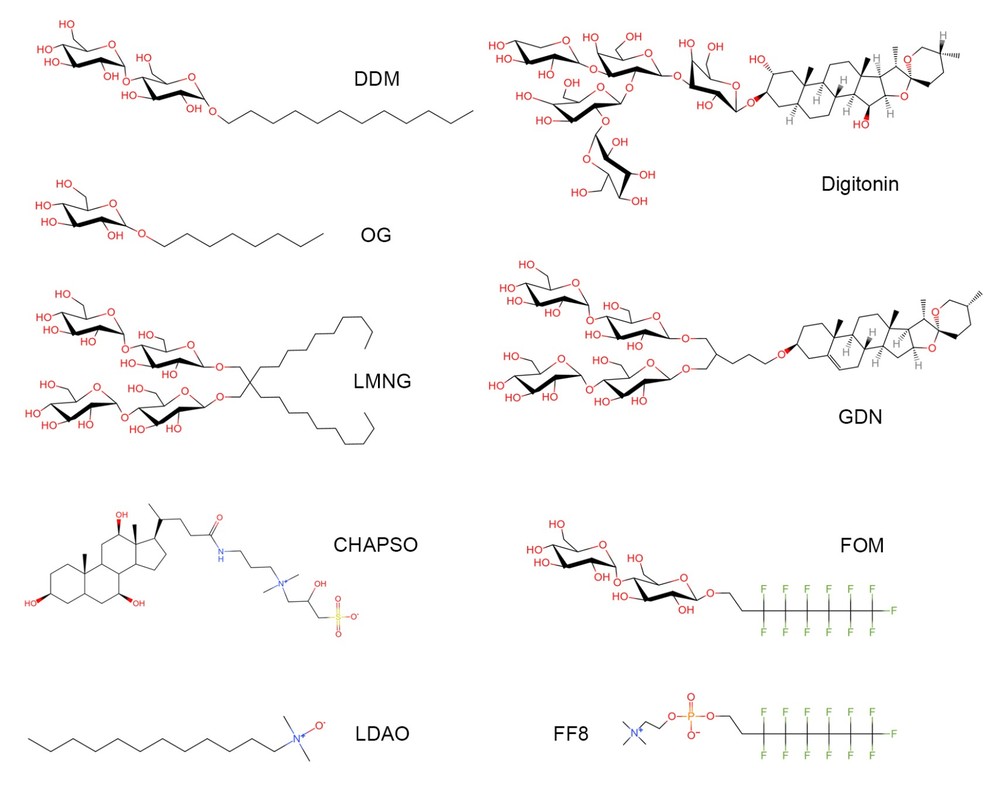 Figure 1. Representative detergent used in structure determination of membrane proteins. (Li, 2022)
Figure 1. Representative detergent used in structure determination of membrane proteins. (Li, 2022)
Longer acyl chain detergents generally offer enhanced stabilization, whereas shorter chain detergents produce smaller micelles that may be advantageous for structural applications. Because protein behavior varies widely, systematic detergent screening remains essential for identifying the optimal balance.
Profacgen has developed a robust and scalable detergent screening platform designed to address the diverse behaviors of membrane proteins from different expression systems. Following cell disruption by homogenization, sonication, or freeze–thaw cycling, membrane fractions are isolated by ultracentrifugation to ensure consistent starting material.
The membrane preparations are then evaluated using a broad panel of detergents representing multiple chemical classes and micelle properties. Our standard detergent library includes:
Each detergent is tested across optimized concentration ranges relative to its critical micelle concentration (CMC). When appropriate, detergent mixtures or stepwise detergent exchanges are evaluated to improve extraction efficiency or enhance protein stability.
By combining chemical diversity with condition optimization, our screening platform enables informed selection of detergents tailored to the protein's structural class, stability requirements, and downstream application goals.
To maximize yield while preserving protein structure and function, we systematically optimize:
This multi-parameter approach allows identification of robust conditions tailored to each target protein.
Each detergent condition is assessed using analytical methods such as SDS-PAGE, Western blotting, and size exclusion chromatography. When required, functional or binding assays are incorporated to confirm activity. The final outcome is a data-driven recommendation for the detergent and conditions best suited for downstream purification and applications.
Background
A pharmaceutical company required highly stable GPCR protein for downstream ligand-binding and signaling assays. However, initial extraction using standard detergents resulted in rapid aggregation and loss of functional activity, making assay results inconsistent and difficult to reproduce.
Our Solution
Profacgen performed a systematic screen of mild non-ionic detergents, focusing on maltoside-based options with different alkyl chain lengths. Detergent concentrations, extraction temperatures, and incubation times were optimized in parallel. In addition, ligand-stabilized extraction buffers were evaluated to enhance conformational stability during solubilization.
Final Results
The optimized detergent system significantly reduced aggregation and preserved native receptor activity. The purified GPCR demonstrated improved stability over time and enabled robust, reproducible ligand-binding assays.
Background
An academic research group aimed to determine the structure of a multi-pass ion channel using cryo-EM but struggled with sample heterogeneity and poor particle distribution due to suboptimal detergent selection.
Our Solution
We screened detergents spanning a range of micelle sizes, including maltosides and advanced neopentyl glycol detergents. Detergent performance was assessed using size-exclusion chromatography and stability analysis to identify conditions that promoted monodispersity. Detergent exchange strategies were also tested to further refine sample quality.
Final Results
An optimal detergent condition was identified that produced homogeneous, stable protein samples. The resulting preparation was well-suited for cryo-EM grid preparation and supported successful high-resolution structural analysis.
Background
A biotechnology company required native transporter protein antigens for antibody discovery but observed epitope degradation and reduced immunoreactivity during detergent-based extraction.
Our Solution
Profacgen combined detergent screening with stabilizing additives and low-temperature extraction protocols to minimize conformational disruption. Both mild detergents and detergent combinations were evaluated to preserve extracellular domains critical for antibody recognition. Extraction conditions were refined to balance solubilization efficiency and structural integrity.
Final Results
The optimized protocol yielded structurally intact transporter proteins with preserved native epitopes. These proteins successfully supported immunization and downstream antibody screening, accelerating the client's therapeutic discovery program.
Reference:
Fill out this form and one of our experts will respond to you within one business day.