E. coli Cell-Free Expression is a rapid and highly versatile in vitro protein production platform that utilizes the transcription and translation machinery extracted from E. coli cells. Unlike conventional cell-based expression, protein synthesis occurs entirely in solution, enabling fast production of recombinant proteins without the constraints of cell growth, viability, or toxicity. Profacgen offers optimized E. coli cell-free expression systems—most notably the S30 extract platform—to support high-yield, parallel synthesis of enzymes, regulatory proteins, membrane proteins, and toxic proteins. This system is ideal for rapid screening, functional validation, and applications requiring precise experimental control and accelerated timelines.
In vitro protein expression, also known as cell-free protein expression, harnesses the native transcriptional and translational machinery of cells while eliminating the complexity of maintaining living hosts. In E. coli cell-free systems, essential components such as ribosomes, tRNAs, amino acids, transcription factors, and metabolic enzymes are retained in cell lysates, enabling efficient protein synthesis upon addition of DNA or mRNA templates.
Compared with cell-based expression, E. coli cell-free platforms allow direct manipulation of reaction conditions, rapid iteration, and efficient expression of proteins that are toxic, unstable, or prone to aggregation in vivo. These characteristics make E. coli cell-free expression a preferred choice for early-stage research, high-throughput screening, and challenging protein targets.
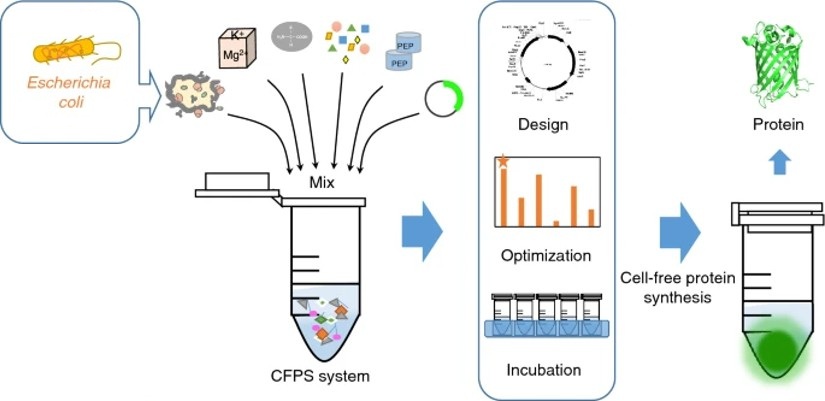 Figure 1. E. coli cell-free expression. (Adapted from Zhang et al., 2021)
Figure 1. E. coli cell-free expression. (Adapted from Zhang et al., 2021)
Among the available E. coli cell-free platforms, the E. coli S30 extract system is the most widely used and robust. It is prepared from protease-deficient E. coli B strains lacking ompT and lon protease activity, significantly reducing protein degradation during synthesis.
Protein expression is initiated by supplementing the S30 extract with amino acids, energy-regenerating substrates, and T7 RNA polymerase. This system supports both transcription and translation in a single reaction and delivers high protein synthesis rates under well-defined conditions.
Profacgen offers end-to-end, highly customizable E. coli cell-free protein expression services designed to support rapid protein production, method development, and difficult-target expression. Each project is tailored based on protein properties, experimental goals, and downstream applications.
Gene & Template Optimization
We perform codon optimization tailored to E. coli cell-free systems, taking into account GC content, rare codons, mRNA secondary structure, and translation efficiency. Clients may provide plasmid DNA or PCR products, or request full template preparation, including promoter and ribosome binding site optimization.
Vector Design & Reaction Setup
Optimized vectors compatible with E. coli S30 extract systems are constructed to ensure robust transcription and translation. Both DNA- and mRNA-driven expression formats are supported, depending on speed and yield requirements.
Rapid Expression Screening & Yield Optimization
Small-scale reactions are used to rapidly screen expression conditions, including magnesium concentration, energy systems, temperature, and reaction formats. This enables quick identification of optimal conditions before scale-up.
Parallel Protein Variant Expression
Multiple constructs—such as mutants, truncations, or tag variants—can be expressed simultaneously in a single workflow, accelerating structure–function studies and lead optimization.
Specialized Protein Production
We support the production of isotope-labeled proteins, fluorescently labeled proteins, and proteins incorporating non-natural or modified amino acids, enabling downstream structural, interaction, or functional studies.
Expression Validation & Quality Assessment
Protein expression is verified at both RNA and protein levels using techniques such as RT-PCR, SDS-PAGE, Western blotting, fluorescence detection, and activity assays.
Overall, Profacgen's E. coli cell-free expression platform provides a fast, flexible, and controllable solution for protein production, especially when traditional cell-based systems are time-consuming or ineffective.

| Step | Common Issues / Variability Sources | Practical Solutions |
|---|---|---|
| Extract Preparation | Inconsistent lysate activity due to growth phase, harvest timing, or cell condition; batch-to-batch extract variability impacting yield. | Standardize culture conditions (growth phase, media, OD at harvest), use optimized lysis protocols (sonication/homogenization), and validate extract activity with control reactions before use. Regularly benchmark new lysate batches. |
| Template Quality | Impure or degraded DNA/RNA templates lower expression efficiency; linear DNA templates are less stable than circular ones. | Ensure high-purity plasmid prep, verify template integrity, incorporate protective elements (e.g., sequence stabilizers) for linear templates, or use circular templates where feasible. |
| Transcription Efficiency | Poor transcription due to suboptimal promoters, template design, or depleted NTPs in reaction mix. | Use validated strong promoters (e.g., T7), optimize untranslated regions (UTRs), and supplement energy systems (NTPs) as needed. |
| Translation Variability | Fluctuating translation rates because of inconsistent magnesium/ion concentrations or ribosome/inhibitor levels in lysate. | Fine-tune reaction buffer (Mg2+, K+), use fresh lysate stocks, and include folding aids or chaperones for sensitive targets. |
| Protein Solubility & Folding | Misfolding or aggregation, especially with membrane or multimeric proteins. | Add detergents, liposomes, or folding partners; adjust temperature and redox environment to favor correct folding. |
| Reaction Stability & Duration | Premature run-down of energy sources limits protein yields. | Use energy-regeneration systems (e.g., creatine kinase/ATP regeneration), continuous exchange formats, or supplement substrates mid-reaction. |
| Batch-to-Batch Variability | Differences in extract lots, reagent quality, or preparation technique lead to inconsistent yields and kinetics. | Maintain strict SOPs for extract prep, use quality-controlled reagents, and include assay controls with every batch. |
| Template Degradation | Endogenous nuclease activity reduces template half-life. | Treat lysate with run-off or dialysis steps to reduce nuclease content and protect templates. |
Client Requirement:
A research group investigating biocatalyst engineering required high-throughput functional comparison of fifteen enzyme variants. Each variant contained subtle active-site mutations hypothesized to improve catalytic efficiency. Traditional E. coli expression necessitated individual cloning, transformation, cultivation, and induction—a workflow spanning several weeks that severely constrained experimental iteration.
Our Solution:
We deployed our E. coli S30 cell-free expression system to eliminate these bottlenecks. Linear expression cassettes were generated directly from PCR amplicons, bypassing cloning entirely. Reactions were assembled in parallel in 96-well format, with optimized magnesium and energy buffer concentrations ensuring uniform expression yields across all variants. No purification was required; clarified lysates were transferred directly to activity assay plates.
Final Results:
All fifteen variants were synthesized, quantified, and kinetically characterized within a single 8-hour workday. The client identified a variant with 4.5-fold improved catalytic efficiency and immediately advanced it to downstream engineering rounds. By compressing months of screening into days, cell-free expression transformed the client's enzyme optimization pipeline and enabled data-driven decisions at unprecedented speed.
Client Requirement:
A client investigating transcriptional regulation required milligram quantities of a human regulatory protein for protein–protein interaction mapping. This factor exhibited profound cytotoxicity when expressed in living E. coli; even minimal leaky expression caused growth arrest, and induction rapidly led to culture collapse. Soluble material was undetectable, and inclusion body refolding proved unsuccessful due to the protein's conformational sensitivity.
Our Solution:
We deployed our E. coli cell-free expression platform to bypass host viability constraints entirely. The linear expression template was amplified directly from the client's plasmid and added to an optimized lysate system. Reaction parameters—including magnesium concentration, redox environment, and incubation temperature—were systematically titrated to favor soluble accumulation over aggregation. Degradation was minimized through chaperone supplementation.
Final Results:
Within three working days, the client received purified, full-length protein at yields sufficient for biochemical interrogation. Surface plasmon resonance confirmed specific binding to its cognate partner, enabling elucidation of a previously inaccessible interaction network. By eliminating the dependency on host viability, cell-free expression provided a direct route to functional protein where cellular systems had repeatedly failed.
Consult Our Experts on Your Project
With extensive expertise in cell-free expression technologies, Profacgen delivers fast, flexible, and reliable E. coli cell-free protein expression solutions. Contact us today to discuss your project and discover how our platform can accelerate your research and development goals.
References:
Fill out this form and one of our experts will respond to you within one business day.