Antibiotic-free expression systems represent a next-generation solution for recombinant protein production, eliminating the need for antibiotic selection while maintaining plasmid stability and high expression efficiency. Traditional expression systems rely heavily on antibiotic resistance genes and selective pressure to retain plasmids during fermentation. However, these approaches introduce regulatory, safety, and environmental concerns due to antibiotic residues and the spread of resistance genes.
At Profacgen, we provide advanced antibiotic-free expression platforms based on toxin–antitoxin stabilization systems, enabling robust plasmid maintenance without antibiotics. Our integrated services cover construct design, cloning, expression optimization, and scalable production. By combining high productivity with regulatory-friendly processes, we deliver high-quality recombinant proteins suitable for research, industrial, and therapeutic applications.
Recombinant protein expression has become a cornerstone of modern biotechnology, supporting applications in pharmaceuticals, diagnostics, industrial enzymes, and basic research. Traditionally, plasmid-based expression systems incorporate antibiotic resistance genes to ensure plasmid retention within host cells during fermentation. Antibiotics are added to the culture medium to eliminate plasmid-free cells, thereby maintaining expression stability.
While effective, this strategy presents several critical limitations:
Given these limitations, the development of antibiotic-free expression systems has become an inevitable trend in modern biomanufacturing.
Among the alternative strategies developed to replace antibiotic selection, toxin–antitoxin (TA) systems have emerged as one of the most effective solutions.
This system operates based on a post-segregational killing mechanism:
When the plasmid is retained within the cell, the antitoxin inhibits the toxin, allowing normal growth and protein expression. However, if the plasmid is lost:
This mechanism ensures stable plasmid maintenance without the need for antibiotics, enabling efficient and safe protein production.
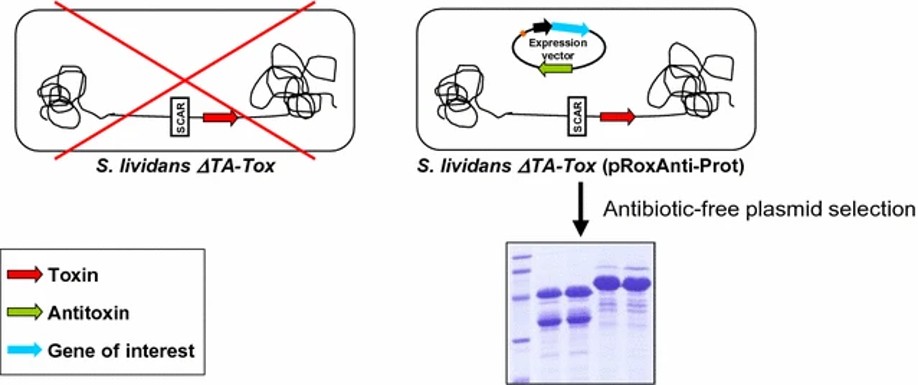 Figure 1. An antibiotic marker-free platform for heterologous protein production in Streptomyces. (Sevillano et al., 2017)
Figure 1. An antibiotic marker-free platform for heterologous protein production in Streptomyces. (Sevillano et al., 2017)
Profacgen provides a comprehensive antibiotic-free protein expression platform, leveraging toxin–antitoxin systems and advanced molecular engineering to deliver stable, high-yield protein production.

Antibiotic-Free Expression Platforms
Our toxin–antitoxin system offers a robust and efficient stabilization mechanism:
Application-Oriented Support
Our antibiotic-free expression systems are suitable for:
Client Requirements:
A research organization aimed to reduce production costs associated with antibiotic use and mandatory removal steps in their recombinant protein manufacturing workflow for non-therapeutic applications.
Our Solution:
Profacgen replaced the traditional antibiotic-based selection system with a toxin–antitoxin platform that maintains plasmid stability without exogenous antibiotics. This simplified the production workflow by eliminating antibiotic addition, monitoring, and removal verification steps while maintaining high expression levels.
Final Results:
Significantly reduced overall production costs through decreased raw material expenses, shortened processing time, and eliminated antibiotic removal validation. Protein quality and yield remained equivalent to or better than antibiotic-based methods. The organization successfully implemented this cost-effective platform across multiple production projects, improving their operational efficiency.
Client Requirements:
A biotech company required production of a therapeutic protein with zero antibiotic contamination to meet stringent regulatory standards for clinical applications. Traditional antibiotic-based selection systems risked carryover of trace antibiotics into final product.
Our Solution:
Profacgen implemented a toxin–antitoxin stabilization system that maintains plasmid retention through essential post-segregational killing mechanisms, completely eliminating the need for antibiotics during fermentation. Expression conditions including media composition, temperature, and induction timing were systematically optimized to maximize yield while maintaining plasmid stability.
Final Results:
Successfully produced high-purity therapeutic protein with no detectable antibiotic residues by sensitive LC-MS analysis, fully meeting regulatory requirements. The antibiotic-free workflow also reduced downstream processing steps and associated costs, accelerating the client's path to clinical trials.
Client Requirements:
A client needed large-scale production of an industrial enzyme with stable expression over extended fermentation periods. Traditional antibiotic-dependent systems risked plasmid loss during prolonged culture, compromising yield and consistency.
Our Solution:
We developed an antibiotic-free expression system incorporating a plasmid stabilization module based on essential gene complementation. This ensured plasmid retention throughout extended high-cell-density fermentation cycles without selective pressure. Process parameters were optimized for industrial scalability.
Final Results:
Achieved consistent, high-yield enzyme production across multiple fermentation batches with no plasmid loss detected. The antibiotic-free approach eliminated contamination risks associated with antibiotic carryover and simplified quality control. The client benefited from improved process efficiency and reduced regulatory burden for their industrial enzyme product.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.