Wheat germ extract (WGE) cell-free protein expression is a versatile and rapid platform for producing functional, full-length proteins without the limitations of living cells. By bypassing cell viability and growth requirements, WGE cell-free systems enable high-yield protein synthesis, cost-effective workflows, and flexible scalability. Profacgen leverages coupled transcription–translation wheat germ extract systems to integrate transcription and translation in a single reaction, simplifying workflows and maximizing protein yield. Our platform supports expression of toxic, membrane, or post-translationally modified proteins, as well as high-throughput applications, making it ideal for research and industrial protein production.
Cell-free protein synthesis (CFPS) systems have been used for decades as a powerful alternative to cell-based expression in both basic research and industrial protein production. Unlike in vivo systems, CFPS eliminates the need for host cell maintenance, cell division, and metabolic balance, thereby removing many bottlenecks associated with toxicity, poor solubility, or slow expression kinetics.
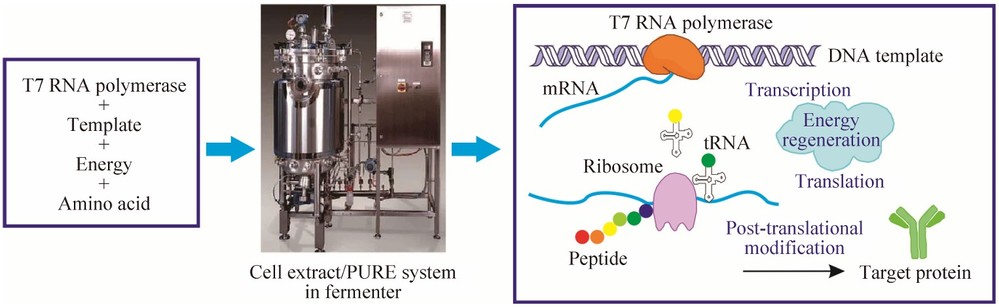 Figure 1. Schematic illustration of industrial production of protein therapeutics using CFPS systems. (Jia et al., 2016)
Figure 1. Schematic illustration of industrial production of protein therapeutics using CFPS systems. (Jia et al., 2016)
Among various CFPS platforms, wheat germ extract systems are particularly advantageous for eukaryotic protein expression. Derived from plant embryos with naturally high translational activity, wheat germ extracts provide a translation machinery that closely resembles eukaryotic intracellular environments. This makes them especially suitable for proteins requiring complex folding, multiple domains, or post-translational features that are often poorly handled by prokaryotic systems.
Traditional wheat germ expression workflows involve two discrete steps: in vitro transcription using SP6, T3, or T7 RNA polymerase, followed by a separate translation reaction. While effective, this approach introduces additional handling steps, time delays, and RNA loss.
To address these limitations, Profacgen employs coupled transcription–translation wheat germ extract systems, in which transcription and translation occur simultaneously within a single reaction vessel. This integrated workflow reduces handling complexity, minimizes RNA degradation, improves reproducibility, and significantly enhances overall protein yield and throughput.
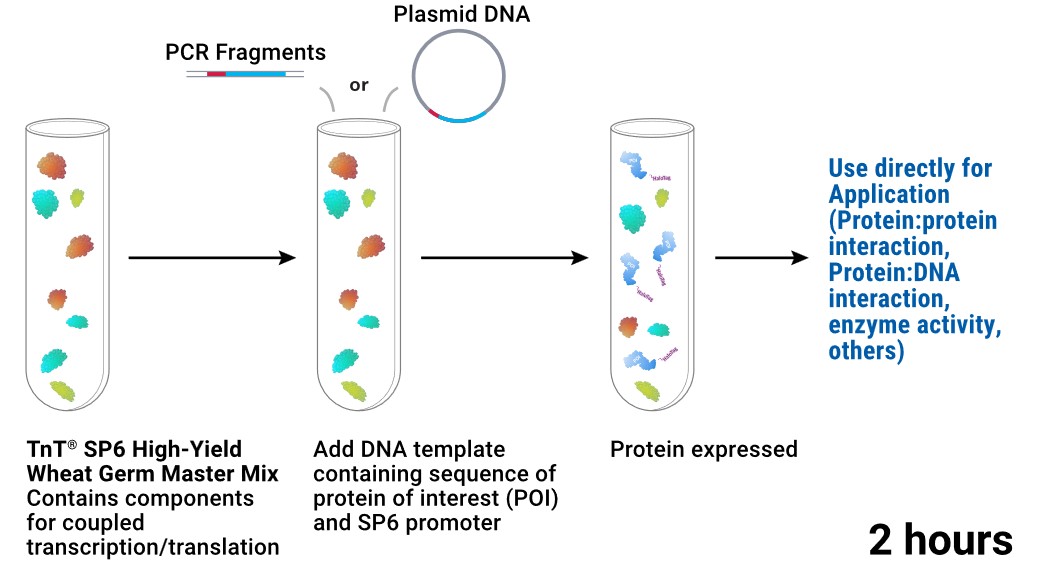 Figure 2. Workflow overview of coupled transcription–translation wheat germ extract cell-free expression systems.
Figure 2. Workflow overview of coupled transcription–translation wheat germ extract cell-free expression systems.
Profacgen provides end-to-end wheat germ extract cell-free protein expression services, tailored to the biochemical characteristics and downstream applications of each target protein.
Custom Protein Expression Services
Single-Tube Coupled Transcription–Translation
Optimized Reaction Components and Protocols
High-Throughput and Parallel Protein Synthesis
Site-Specific Labeling and Unnatural Amino Acid Incorporation
Protein Purification and Downstream Processing
| Steps | Details |
|---|---|
| Step 1. Reaction Component Preparation | Profacgen prepares highly active wheat germ extract containing endogenous ribosomes, tRNAs, and all essential translation factors (initiation, elongation, and termination factors). To support specialized labeling or functional studies, three optimized amino-acid master mixes are available:
|
| Step 2. In vitro Transcription | Plasmid DNA or PCR-amplified templates encoding the target gene are added to the transcription reaction. Under the action of the selected RNA polymerase, high-quality mRNA is synthesized in vitro and directly enters the translation phase, minimizing handling steps and template loss. |
| Step 3. Cell-Free Translation | The synthesized mRNA is translated using the wheat germ extract in the presence of amino acids, energy substrates, and cofactors. This eukaryotic translation system provides a favorable environment for proper folding and functional expression of complex proteins. |
| Step 4. Protein Detection and Purification | Protein expression is monitored using appropriate analytical methods (e.g., fluorescence, enzymatic activity, or immunodetection). The target protein can then be purified using tag-based or tag-free purification strategies, depending on project requirements. |
| Step 5. mRNA Recycling and Reaction Extension | After translation, released mRNA can be reused for additional translation cycles within the same system, increasing overall protein yield. Over multiple cycles, mRNA is gradually inactivated, ensuring controlled reaction termination and reproducibility. |
Client Requirement:
A biotechnology company required rapid production of a full-length serine/threonine kinase for biochemical characterization and inhibitor screening. The target protein showed severe cytotoxicity in E. coli and repeated attempts using bacterial and insect cell systems resulted in low yield and extensive truncation. Time constraints ruled out lengthy cell-line optimization.
Our Solution:
Profacgen applied the coupled transcription–translation wheat germ extract cell-free expression system, enabling simultaneous transcription and translation in a single reaction. Reaction conditions, amino acid composition, and energy regeneration were optimized to support efficient synthesis and proper folding of the kinase.
Final Results:
Within 24 hours, milligram-scale quantities of soluble, full-length kinase were obtained. Enzymatic assays confirmed robust catalytic activity, allowing immediate use in inhibitor screening without additional refolding steps.
Client Requirement:
An academic research group needed to express a panel of membrane protein mutants for structure–function analysis. Conventional cell-based expression systems produced inconsistent yields and required extensive optimization for each variant, significantly delaying the project timeline.
Our Solution:
We implemented a high-throughput wheat germ cell-free expression workflow in a microplate format. Expression conditions were standardized across all variants, with targeted adjustments to reaction composition and solubilization strategies to enhance membrane protein stability and folding.
Final Results:
All target variants were successfully expressed in parallel with high reproducibility. More than 80% of the proteins were obtained in soluble form and were immediately compatible with downstream biochemical and binding studies, significantly accelerating the client's research progress.
Consult Our Experts on Your Project
Reference:
Fill out this form and one of our experts will respond to you within one business day.