The Microalgae Expression System represents a sustainable, animal-free, and highly scalable platform for recombinant protein production, offering a compelling alternative to traditional microbial, insect, and mammalian expression systems. Leveraging fast-growing, photosynthetic microalgae such as Chlamydomonas reinhardtii, this platform enables cost-effective production of recombinant proteins with essential eukaryotic post-translational modifications. Microalgae are free from human and animal pathogens, require minimal nutritional input, and can be cultivated at large scale using simple infrastructure. Profacgen has established a comprehensive microalgae-based expression platform that supports gene design, transformation, expression optimization, and downstream purification, delivering high-quality proteins for research, diagnostic, and preclinical applications.
Over the past several decades, transgenic plant-based expression systems have attracted increasing attention from the biopharmaceutical industry. Among these systems, microalgae—also referred to as microphytes—have emerged as particularly attractive hosts for recombinant protein production. Microalgae comprise a diverse, polyphyletic group of organisms ranging from prokaryotes (e.g., Synechococcus) to eukaryotes (e.g., Chlamydomonas), many of which have long histories of safe use in food, feed, nutraceuticals, and cosmetic products.
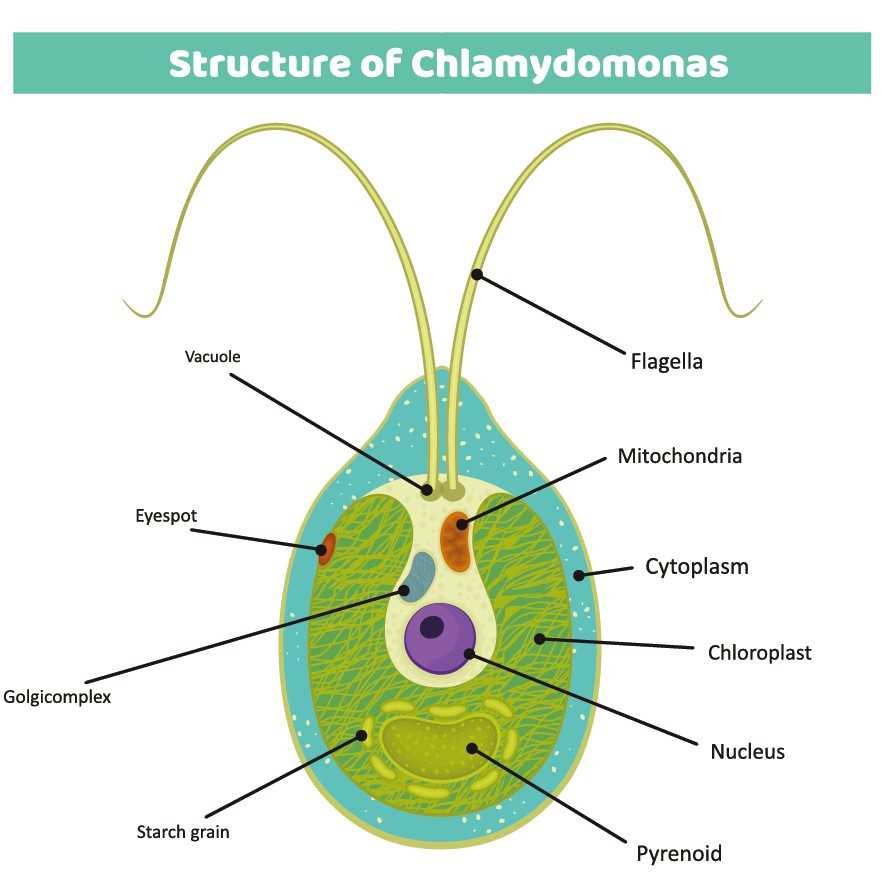
Unlike higher plants, microalgae grow rapidly, often doubling their biomass within hours, and can be cultivated year-round in controlled photobioreactors or open systems. As solar-powered biofactories, they require only light, carbon dioxide, water, and simple inorganic nutrients, resulting in low production costs and minimal environmental footprint. Importantly, many microalgal species are generally regarded as safe (GRAS), making them particularly suitable for applications related to human and animal health.
From a biopharmaceutical perspective, eukaryotic microalgae offer a critical advantage over bacterial hosts: the ability to perform post-translational modifications such as disulfide bond formation and glycosylation, which are often required for proper protein folding, stability, and biological activity. At the same time, microalgae are toxin-free and free of mammalian pathogens, significantly enhancing biosafety and reducing downstream purification burdens. These features position microalgae as a promising bridge between low-cost microbial systems and high-fidelity but expensive mammalian cell platforms.
Among the many microalgal species explored for recombinant protein production, Chlamydomonas reinhardtii stands out as the most extensively studied and widely adopted model organism. This unicellular green alga is genetically tractable, grows rapidly, and is supported by a rich toolbox of molecular biology resources.
A unique advantage of C. reinhardtii is the ability to express recombinant proteins from either the nuclear genome or the chloroplast genome. Nuclear expression allows secretion or intracellular targeting of proteins and supports certain post-translational modifications, while chloroplast expression enables exceptionally high protein accumulation and genetic stability without gene silencing. This dual-genome flexibility allows expression strategies to be tailored to the structural and functional requirements of each target protein.
Over the past decades, C. reinhardtii has been successfully used to produce enzymes, antigenic peptides, vaccine candidates, antibody fragments, and industrial proteins. As interest in sustainable and animal-free biomanufacturing continues to grow, this algal system is increasingly recognized as a valuable platform for next-generation biopharmaceutical development.
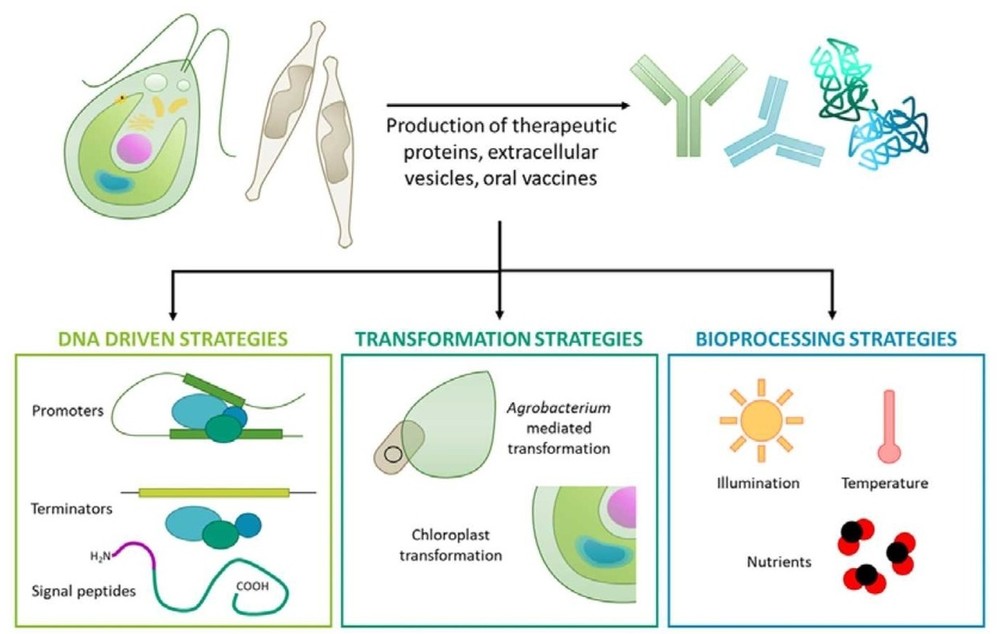 Figure 1. Production of recombinant and therapeutic proteins in microalgae. (Banerjee and Ward, 2022)
Figure 1. Production of recombinant and therapeutic proteins in microalgae. (Banerjee and Ward, 2022)
Profacgen provides a one-stop recombinant protein production service based on the Chlamydomonas reinhardtii expression system. Our services span the entire workflow, from gene design to protein purification, and are tailored to meet diverse client requirements.
| Service | Details |
|---|---|
| Gene Design and Codon Optimization | We perform rational gene design optimized specifically for microalgal expression. Special attention is given to codon bias differences between nuclear and chloroplast genomes, ensuring efficient translation in the chosen expression compartment. Optional fusion tags, targeting signals, and reporter elements can be incorporated to enhance expression, localization, or purification. |
| Promoter and Regulatory Element Selection | Expression efficiency in microalgae is strongly influenced by promoter and untranslated region (UTR) selection. Profacgen employs a range of well-characterized regulatory elements, including optimized chloroplast promoters and UTRs, fusion strategies such as the 16S ribosomal promoter with the atpA 5' UTR, and nuclear promoter combinations like hsp70 fused with rbcs2. These strategies enable robust and controllable transgene expression. |
| Transformation and Strain Engineering | We offer both nuclear and chloroplast transformation using validated protocols. Selectable markers such as humanized enhanced GFP (eGFP) and other reporter systems are employed to facilitate rapid screening and identification of high-expressing clones. Stable transformants are characterized to ensure genetic integrity and expression consistency. |
| Cultivation and Expression Optimization | Microalgae cultivation parameters—including light intensity, temperature, nutrient composition, and induction conditions—are systematically optimized to maximize biomass accumulation and protein yield. Our scalable cultivation platforms support both small-scale feasibility studies and larger-scale production campaigns. |
| Protein Extraction and Purification | Following expression, algal biomass is harvested and processed using optimized lysis and extraction methods designed to preserve protein integrity. We provide a range of purification options, including affinity chromatography, ion exchange, and size-exclusion chromatography, delivering proteins that meet defined purity and activity specifications. |
Background
A biotechnology company developing industrial biocatalysts required milligram quantities of a eukaryotic enzyme for substrate specificity screening. The protein required disulfide bond formation and chaperone-assisted folding unavailable in E. coli, yet mammalian production costs were prohibitive for early-stage screening campaigns.
Our Solution
We engineered Chlamydomonas reinhardtii for nuclear expression using a codon-optimized synthetic gene. A strong endogenous promoter drove transcription, and an N-terminal fusion partner enhanced soluble accumulation. Cultivation parameters—including light intensity, photoperiod, and carbon dioxide supplementation—were systematically optimized to maximize volumetric productivity while maintaining protein quality.
Final Results
The client received multiple milligrams of purified, catalytically active enzyme with confirmed batch consistency. The material enabled successful screening of thirty substrate candidates, leading to identification of a promising lead variant for further engineering. By accessing eukaryotic folding machinery at algal production costs, we bridged the gap between bacterial incompatibility and mammalian expense, allowing the client to advance their biocatalyst pipeline with minimal upfront investment.
Background
A research institute investigating immune signaling pathways required a recombinant antibody fragment for surface plasmon resonance binding studies. Strict animal-free policies prohibited hybridoma generation or mammalian cell culture, and bacterial expression yielded insoluble, non-functional aggregates.
Our Solution
We deployed a streamlined nuclear expression workflow in Chlamydomonas reinhardtii. The single-chain variable fragment was fused to an endogenous secretion signal, directing the protein to the culture medium and eliminating mechanical disruption requirements. Affinity purification from conditioned medium delivered high-purity material within two weeks of construct transformation.
Final Results
The purified antibody fragment retained sub-nanomolar binding affinity to its target antigen, confirmed by independent SPR analysis. Thermal shift assays demonstrated excellent stability across multiple freeze-thaw cycles. This case validated microalgae as a viable, regulatory-alternative host for recombinant antibody production, combining the speed of microbial systems with eukaryotic folding capacity. The client now routinely employs algal-derived fragments for reagent generation, bypassing animal use entirely.
Consult Our Experts on Your Project
Profacgen provides comprehensive Microalgae Expression System services built on proven expertise in Chlamydomonas reinhardtii. From gene design to purified protein, we deliver flexible, cost-effective, and animal-free solutions tailored to your project goals. Contact us to discuss how our microalgae platform can support your next recombinant protein program.
Reference:
Fill out this form and one of our experts will respond to you within one business day.