Peptide or compound synthesis is the crucial step of design for E3 ligase or target protein in protein degradation assay. Peptide synthesis mostly often occurs by coupling the carboxyl group of the incoming amino acid to the N-terminus of the growing peptide chain (C-to-N), which is opposite from protein biosynthesis. Though common methods of peptide synthesis have some critical differences, they all follow the same precise, step-wise and cyclic method to add amino acids one-at-a-time to the growing peptide chain.
Solid phase peptide synthesis (SPPS) is a widely recognized technology in chemistry among biologists for its high-speed, versatility, and automatization possibility. In SPPS system peptide is anchored to an insoluble and stable support (resin), chemical reactants can freely diffuse into the interior of the resin beads, and the reaction rates are close to those in solution. The peptide is assembled in a stepwise manner by repeated cycles of formation of a peptide bond between the amino group and an Nα-protected amino acid, followed by deprotection of the temporary Nα-protecting group. At the end of the synthesis, the assembled peptide is cleaved from the solid support. Figure 1 shows the SPSS process, where Y is a temporary protective group, the side chains of groups Rn are protected in accordance with their functionality.
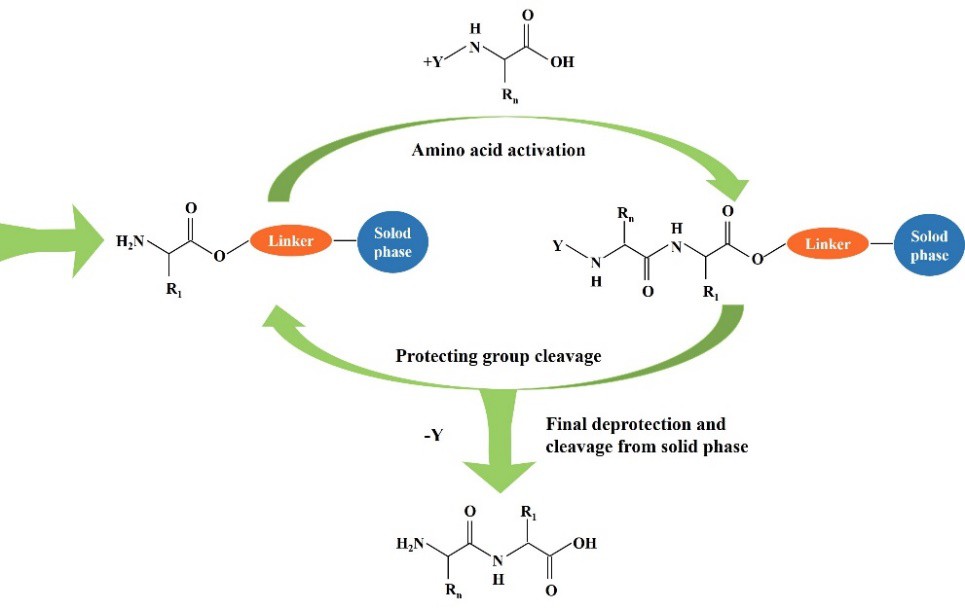 Figure 1. General scheme of SPPS.
Figure 1. General scheme of SPPS.
Profacgen provides specific peptides and new chemical compounds with our professional scientific team and stable platforms to meet different needs including purity levels, modifications and formats. Based on our proven solid-phase peptide synthesis technology, customers can customize 100% accurate peptides of 2-150 amino acids, and purities from crude to >98%. Besides, we provide comprehensive raw data and analytical data as documentation with custom peptides. We test and validate each custom peptide at each stage via LC-MS, MALDI-MS or HPLC to ensure its fidelity.
Profacgen has accumulated lots of experience in custom peptide and compound synthesis. Our professional technical team can provide customers with high-quality peptide synthesis services and many related featured services you may be interested in. Our competitive prices and extensive expertise have earned us the trust of our collaborators. Contact us to find out how Profacgen could be of assistance.
Fill out this form and one of our experts will respond to you within one business day.