Protein engineering is a cornerstone of modern biotechnology, enabling the rational design and optimization of proteins for enhanced performance, novel functionality, and improved therapeutic potential. By combining genetic modification, structural design, and evolutionary strategies, protein engineering allows precise control over protein properties such as stability, activity, specificity, and expression efficiency.
At Profacgen, we offer a comprehensive protein engineering platform that integrates mutagenesis, directed evolution, structural optimization, and advanced functional validation. Our services support a wide range of applications, including enzyme engineering, therapeutic protein development, vaccine design, and fundamental biological research. With a strong foundation in both rational design and high-throughput screening technologies, we provide end-to-end solutions tailored to meet the diverse needs of academic and industrial clients.
Proteins are central to nearly all biological processes, acting as enzymes, signaling molecules, structural components, and therapeutic agents. However, naturally occurring proteins are not always optimized for industrial, medical, or research applications. Protein engineering addresses this limitation by enabling the modification of protein sequences and structures to achieve desired properties.
Key objectives of protein engineering include:
These capabilities have made protein engineering indispensable in fields such as drug discovery, diagnostics, synthetic biology, and industrial biotechnology.
Protein engineering approaches can be broadly categorized into three main strategies:
Rational Design
Rational design involves the use of structural and functional knowledge to introduce targeted modifications. This approach relies on:
Rational design is particularly effective when detailed structural information is available.
Directed Evolution
Directed evolution mimics natural selection in the laboratory by generating diverse protein variants and selecting those with improved properties. Techniques include:
Advanced methods such as continuous evolution systems further accelerate this process.
Hybrid Approaches
In practice, protein engineering often combines rational design and directed evolution to achieve optimal results. Initial rational modifications can guide library design, while iterative screening refines performance.
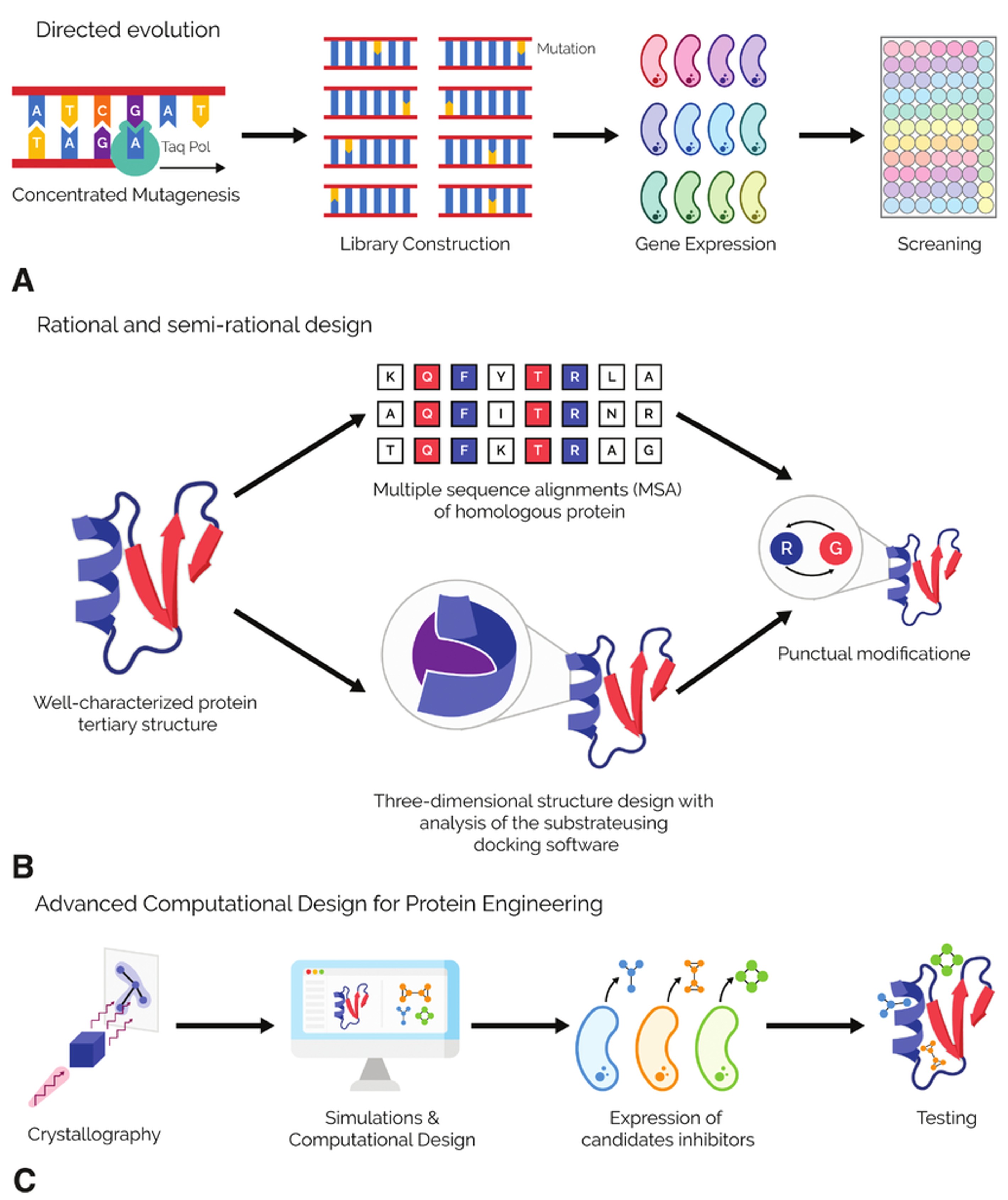
Figure 1. Protein engineering approach. (A) Directed evolution. (B) Rational and semirational design. (C) Advanced computational design for protein engineering. (Otun et al., 2023)
Recent advances have significantly broadened the scope of protein engineering:
These innovations enable the development of increasingly sophisticated protein-based solutions across multiple industries.
Profacgen provides a full spectrum of protein engineering services, covering all major strategies and applications. Our platform is designed to be modular and scalable, allowing seamless integration of multiple approaches within a single project.
| Services | Details |
|---|---|
| Protein Mutagenesis and Structural Optimization | We offer comprehensive mutagenesis and structural engineering services to explore and refine protein function:
|
| Directed Evolution Platforms | Our directed evolution services support the rapid optimization of proteins under defined selection pressures:
We provide cutting-edge continuous evolution systems, including:
|
| Protein Degrader Development | We support the design and optimization of targeted protein degradation systems:
|

Challenge:
A client required an enzyme with improved thermal stability and catalytic efficiency for industrial applications. The wild-type enzyme exhibited insufficient performance under process conditions, limiting throughput and increasing operational costs.
Approach:
Profacgen employed a hybrid protein engineering strategy combining rational design with directed evolution. Computational modeling identified regions amenable to stabilizing mutations, while iterative rounds of random mutagenesis generated diverse variant libraries. High-throughput screening was conducted under elevated temperature conditions to selectively enrich for variants with enhanced thermal tolerance and catalytic activity.
Outcome:
Identified enzyme variants demonstrating significantly improved thermal stability and catalytic efficiency compared to the wild-type enzyme. The optimized variants maintained robust activity under demanding industrial processing conditions, enabling successful scale-up. The client achieved cost-effective manufacturing with enhanced productivity, facilitating commercial deployment of the engineered enzyme.
Challenge:
A biotech company aimed to degrade a disease-associated protein that was previously considered undruggable using conventional inhibitor approaches. Selective elimination of this target offered a promising therapeutic strategy.
Approach:
Profacgen designed a series of heterobifunctional degrader molecules incorporating target-binding warheads and E3 ligase-recruiting ligands. Linker composition and geometry were systematically optimized to facilitate stable ternary complex formation. Degradation activity was validated in cell-based assays across multiple disease-relevant cell lines to assess potency and selectivity.
Outcome:
Achieved efficient and selective degradation of the disease-associated protein with nanomolar potency. The lead degrader candidate demonstrated robust target knockdown and favorable preliminary pharmacokinetic properties. This validated strategy advanced a promising therapeutic candidate for further preclinical development and optimization.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.