The Baculovirus Expression Vector System (BEVS) is a powerful tool for recombinant protein production in insect cells, but its application in mammalian cells has historically been limited due to low yields. Profacgen's BacMam system introduces mammalian-active expression cassettes into baculoviruses, enabling efficient, transient gene expression across multiple mammalian cell lines, including CHO and HEK293 cells. This platform provides high protein yield, broad cell tropism, and flexible transduction, allowing rapid expression of proteins that are difficult to produce via conventional lipid-mediated transfection or other mammalian systems. BacMam is ideal for research, diagnostics, and preclinical applications requiring complex proteins.
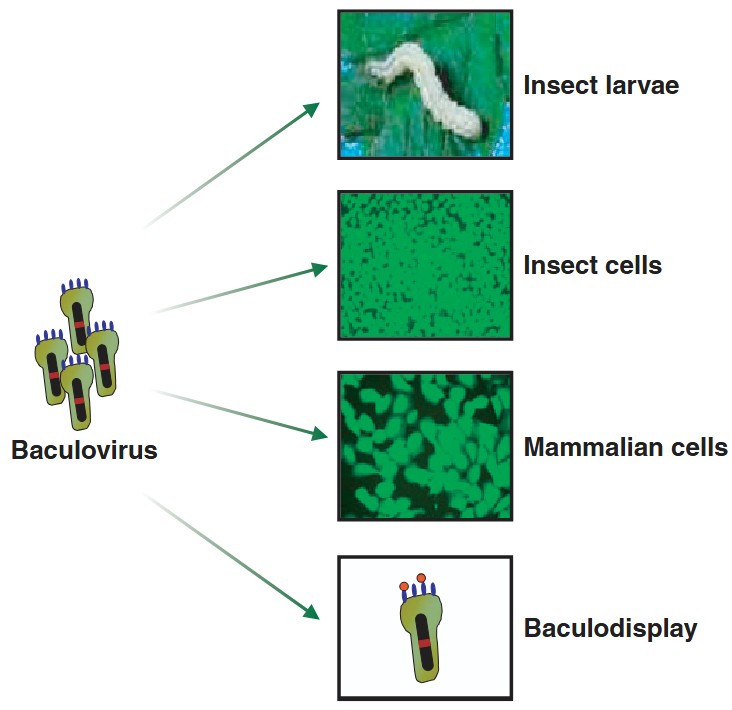 Figure 1. Versatility of baculovirus expression vectors.
Figure 1. Versatility of baculovirus expression vectors.
(Kost et al., 2005)
Traditional BEVS is optimized for insect cells, which limits its direct utility in mammalian protein production. Mammalian cells, on the other hand, provide correct post-translational modifications, proper folding, and biologically relevant glycosylation patterns. However, standard lipid-mediated transient transfection in mammalian cells often achieves only 0.5–10 mg/L protein yield, which may be insufficient for low-abundance or complex proteins.
Profacgen's BacMam system overcomes these limitations by incorporating mammalian-driven promoters into baculoviruses, enabling high-efficiency gene delivery and protein expression in mammalian hosts. This approach supports transient expression in a wide variety of cell types, reduces optimization time, and increases yield to >10 mg/L, making it suitable for challenging targets, low-abundance proteins, and multi-domain or structurally complex proteins.
Key advantages of BacMam over conventional transfection include:
Profacgen provides comprehensive BacMam services tailored to research, diagnostic, and preclinical applications. Our offerings include:
| Service Component | Description |
|---|---|
| BacMam Plasmid Design & Virus Generation |
|
| Mammalian Cell Transduction & Expression Optimization |
|
| Protein Production & Purification |
|
| Stable Cell Line Development (Optional) |
|
| Analytical Characterization |
|
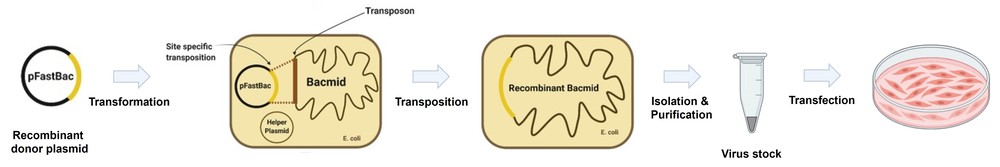
Background
A research institute needed substantial quantities of a low-abundance human cytokine for preclinical activity assays. The protein possessed complex disulfide pairing and was notoriously difficult to produce; conventional lipid-based transfection of HEK293 cells delivered only trace amounts, insufficient for downstream evaluation.
Our Solution
We implemented a BacMam solution by engineering a recombinant baculovirus carrying the cytokine gene under a potent mammalian expression cassette. To overcome poor transduction, we systematically optimized infection parameters—titrating multiplicity of infection, modulating temperature shifts, and applying repeated viral additions to boost gene delivery efficiency.
Final Results
This approach yielded over 12 mg/L of properly folded cytokine, representing a dramatic improvement over transient transfection. Purification proceeded without complication, and both ELISA-based quantification and cell-based activity assays confirmed full biological function. The client received high-quality protein ready for preclinical dosing studies, bypassing lengthy cell line development and avoiding project delays.
Background
A biotech company required a multi-domain human metalloenzyme for crystallography screening and kinetic characterization. Transient transfection in suspension CHO cells generated predominantly misfolded aggregates; soluble yields fell well below the threshold required for structural trials.
Our Solution
We deployed a BacMam platform featuring a bicistronic design to facilitate detection and streamline clone selection. Multiple native and engineered secretion leaders were screened to identify optimal signal peptides for this particular enzyme. Transduction efficiency was enhanced through phosphate-buffered saline dilution of viral stocks and extended low-temperature incubation post-infection.
Final Results
Final expression levels exceeded 11 mg/L of correctly processed, catalytically active enzyme. Size-exclusion chromatography confirmed monodispersity suitable for crystallization trials. The client successfully obtained diffracting crystals within months rather than years, dramatically reducing both the cost and timeline typically associated with mammalian structural biology projects.
Background
A vaccine developer needed a mammalian-expressed viral envelope protein bearing authentic post-translational modifications. Preserving native glycosylation patterns and conformational epitopes was essential for immunogenicity studies, yet stable cell line generation was prohibitively slow for their accelerated timeline.
Our Solution
We constructed a BacMam vector incorporating a strong mammalian promoter and an efficient secretion signal, then transduced HEK293 cells under optimized conditions. Targeted media additives and repeated viral dosing strategies were employed to maximize volumetric productivity while maintaining stringent quality control over protein processing.
Final Results
The campaign delivered approximately 12 mg/L of purified antigen with uniform, complex-type glycosylation closely matching the native viral protein. Correct folding was confirmed through conformation-specific antibody binding, and subsequent mouse immunizations elicited robust neutralizing titers. The client advanced directly to preclinical efficacy studies without the expense and delay of developing stable producer lines.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.