Although small molecule-based crosslinkers are economic and efficient for conjugation chemistry, they may not be the best fit for some applications such as food manufacturing and pharmaceutical production, where residual organic compounds cannot be tolerated and may lead to safety incidents. Such concerns, however, can be largely addressed by our selection of enzyme-based crosslinking strategies1 as they inherently have high substrate specificity. Enzymes, as their structural integrity and function is best preserved under physiological conditions, are expected to be more favorable in crosslinking proteins than chemical approaches that often require unusual pH conditions or the use of organic solvent. Therefore, enzymatic crosslinking strategies not only provide site-specific reaction but also deliver maximized preservation of the structural integrity of proteins.
At Profacgen, a team of experienced biochemists provide enzymatic crosslinking for each specific research request. These enzyme-based approaches present several unique advantages:
Profacgen has established a diverse range of enzymatic crosslinking methods, including:
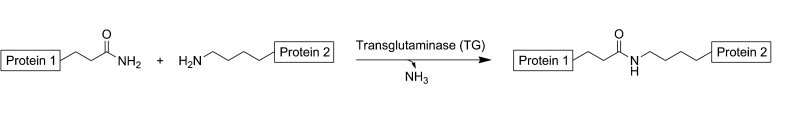
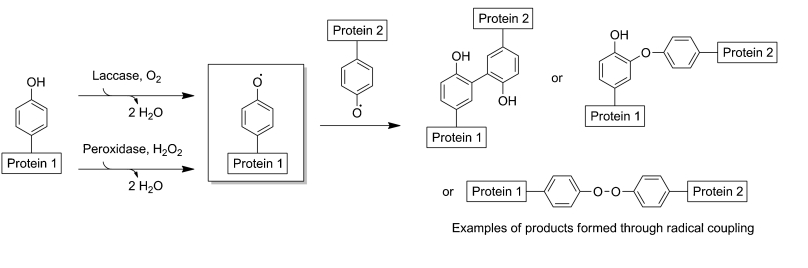
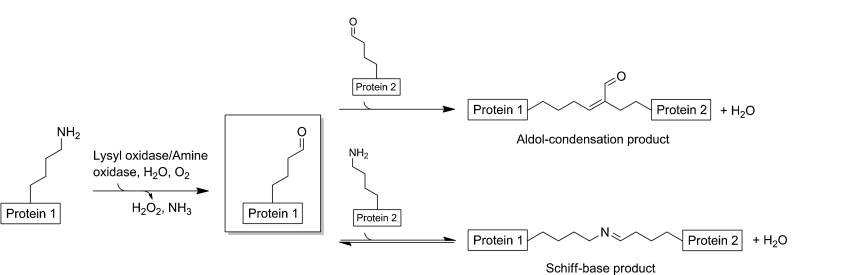

The development of enzyme-based approaches for site-specific crosslinking between proteins has seen a surge in recent years. Two most advantages for enzymatic crosslinking are: First, enzymes usually work in conditions that are most optimal for proteins to display their functions. Two, the natural selectivity of enzymes can be exploited to offer crosslinks in any region or site in the protein molecule. Profacgen offers a wide range of enzymatic strategies for protein crosslinking either by our own state-of-the-art laboratory or through our industry-leading collaborators.
For more information regarding Profacgen’s protein crosslinking service, please contact us. Our customer service representatives are available 24 hours a day, Monday through Friday, to assist you.
References:
1. Heck, T.; et al. Enzyme-catalyzed protein crosslinking. Applied Microbiology and Biotechnology. 2013, 97(2): 461-475.
Fill out this form and one of our experts will respond to you within one business day.