Nanodisc-based protein purification is an advanced, detergent-free or detergent-minimized strategy designed for membrane proteins that are unstable, inactive, or degraded under conventional solubilization conditions. By reconstituting membrane proteins into nanoscale lipid bilayers stabilized by membrane scaffold proteins (MSPs), nanodiscs provide a native-like membrane environment that preserves protein conformation, dynamics, and biological activity. Profacgen offers comprehensive nanodisc-based purification solutions integrated with cell-free expression, direct membrane solubilization, and post-purification reconstitution workflows. Our platform enables reliable isolation of detergent-sensitive, low-abundance, or structurally complex membrane proteins for functional studies, antibody discovery, and structural biology applications.
Membrane proteins are embedded in lipid bilayers and stabilized by extensive hydrophobic interactions. Traditional purification methods rely heavily on detergents to extract these proteins into aqueous solutions. While detergents are effective for many targets, a subset of membrane proteins is highly detergent-sensitive, leading to:
These challenges are particularly pronounced for multi-pass transmembrane proteins, receptors, ion channels, and protein–lipid complexes, where lipid interactions are essential for stability and function.
Nanodiscs are nanoscale, discoidal lipid bilayers stabilized by amphipathic membrane scaffold proteins (MSPs) derived from apolipoprotein A-I. A typical nanodisc consists of:
Structurally, nanodiscs resemble pie-shaped high-density lipoprotein (HDL) particles in vivo, providing a soluble yet membrane-like environment.
Unlike detergent micelles, nanodiscs:
For many applications, a ~10 nm planar lipid bilayer is ideal, as it:
Nanodiscs have been widely used in cryo-EM, NMR, SPR, BLI, enzymatic assays, and antibody screening, enabling high-resolution insights into membrane protein structure and function.
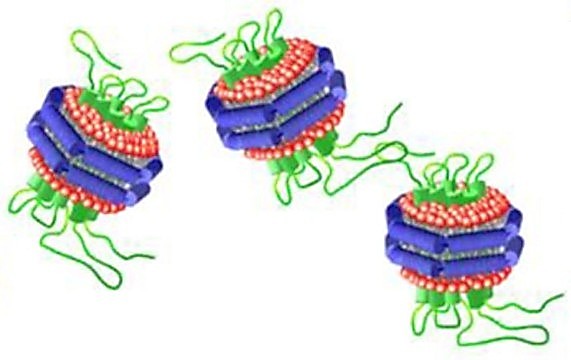
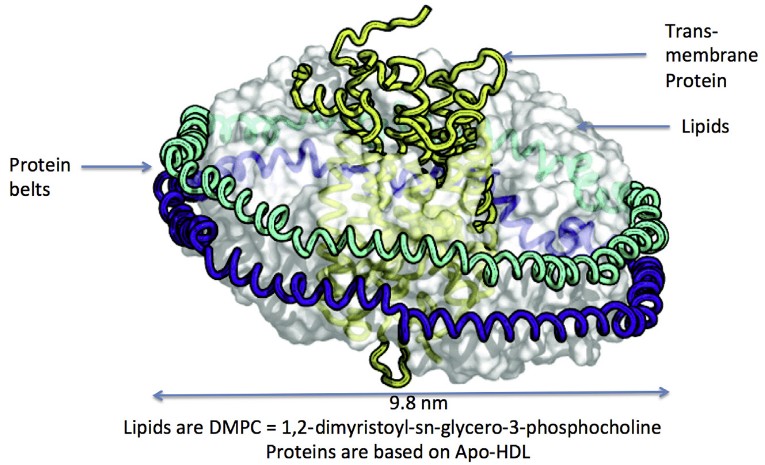 Figure 1. Schematic drawing of a protein-belted nanodisc. (Rosenberg et al., 2019)
Figure 1. Schematic drawing of a protein-belted nanodisc. (Rosenberg et al., 2019)
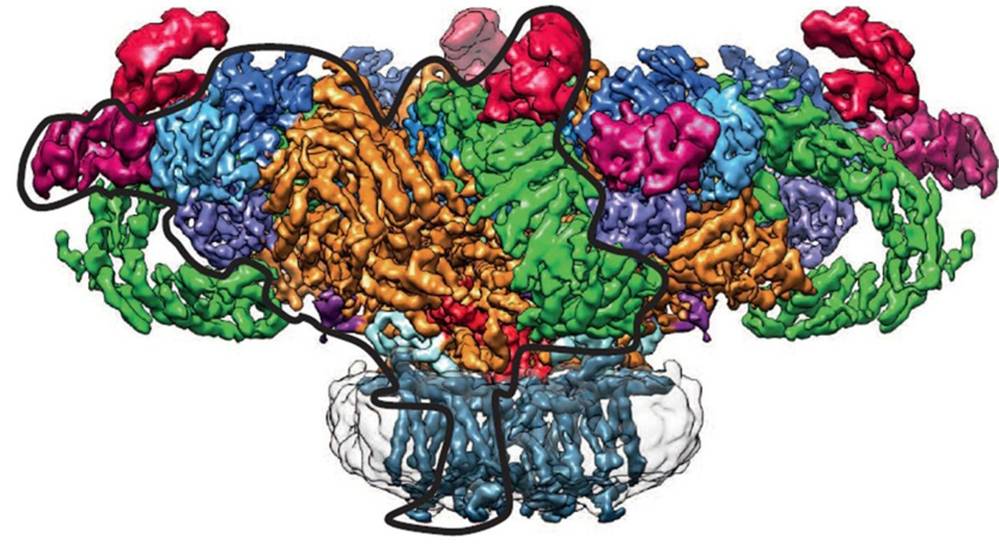 Figure 2. Structure of the ryanodine receptor reconstituted in a nanodisc and resolved to ~6.1 Å by cryo-EM (Denisov and Sligar, 2016).
Figure 2. Structure of the ryanodine receptor reconstituted in a nanodisc and resolved to ~6.1 Å by cryo-EM (Denisov and Sligar, 2016).
Profacgen has established a robust and flexible nanodisc technology platform designed to address the purification and stabilization of detergent-sensitive and structurally complex membrane proteins. Our services are modular and customizable, allowing seamless integration with upstream expression systems and downstream analytical workflows.
| Strategy | Best Suited For | Workflow Overview | Key Features / Advantages | Typical Applications |
|---|---|---|---|---|
| Nanodisc Integration with Cell-Free Expression Systems | Highly toxic membrane proteins; unstable targets; isotope-labeled proteins; rapid screening projects | Preassembled nanodiscs are added directly to a cell-free expression system. During protein synthesis, nascent membrane proteins are co-translationally inserted into the nanodisc bilayer without detergent exposure. |
|
Structural biology (cryo-EM, NMR), mechanistic studies, functional screening of difficult targets |
| Direct Solubilization from Native Membranes into Nanodiscs | Proteins expressed in cells but unstable in detergent alone | Detergents, membrane scaffold proteins (MSPs), and phospholipids are added directly to native membranes. Target proteins, native lipids, and MSPs self-assemble into nanodisc complexes during solubilization. |
|
Functional assays, antibody discovery, biochemical characterization |
| Reconstitution of Detergent-Solubilized Proteins into Nanodiscs | Previously purified membrane proteins requiring stabilization or functional recovery | Detergent-purified proteins are mixed with optimized phospholipids and MSP variants. Controlled detergent removal drives spontaneous nanodisc assembly around the target protein. |
|
Cryo-EM sample preparation, ligand-binding assays, protein–protein and protein–lipid interaction studies |
| Nanodisc Design and Optimization Services | Projects requiring precise control of protein state or lipid environment | Systematic optimization of lipid composition, MSP variants, protein-to-nanodisc ratios, and oligomeric state to meet specific research goals. |
|
Advanced structural studies, mechanistic research, assay development |
Background
A pharmaceutical client needed a functionally active GPCR antigen for antibody discovery and affinity screening. Previous attempts using mild detergents resulted in rapid protein degradation, loss of ligand-binding activity, and poor reproducibility, making the protein unsuitable for SPR and immunization studies.
Our Solution
We implemented a direct membrane solubilization strategy followed by nanodisc assembly using optimized MSP variants and cholesterol-enriched phospholipids. Detergent exposure was minimized and carefully removed during nanodisc formation to preserve native conformation. Lipid composition and protein-to-nanodisc ratios were systematically optimized.
Final Results
The GPCR was purified as a homogeneous, monodisperse nanodisc complex, retaining over 80% ligand-binding activity. The final product supported robust SPR analysis and antibody screening workflows, enabling successful downstream discovery efforts.
Background
A research group sought to produce a multi-pass ion channel for functional and structural studies. Expression in bacterial and mammalian cells consistently failed due to severe cytotoxicity and extremely low yields, preventing purification and characterization.
Our Solution
We integrated preassembled nanodiscs directly into a cell-free expression system, enabling co-translational insertion of the ion channel into a lipid bilayer without the use of detergents. Nanodisc size, lipid composition, and reaction conditions were optimized to favor correct folding and membrane insertion.
Final Results
Milligram-scale quantities of stable, functional ion channel–nanodisc complexes were obtained. The protein demonstrated expected activity in downstream electrophysiological assays and showed excellent behavior in cryo-EM grid preparation, supporting high-quality structural analysis.
Background
A biotechnology company was preparing a membrane transporter for cryo-EM structure determination. Although detergent-based purification yielded sufficient protein quantities, the sample showed aggregation, heterogeneity, and poor particle distribution on cryo-EM grids.
Our Solution
We reconstituted the detergent-purified transporter into nanodiscs using a tailored lipid mixture designed to mimic its native membrane environment. Protein-to-nanodisc ratios, MSP variants, and detergent removal conditions were optimized to promote monodispersity and structural stability.
Final Results
The nanodisc-reconstituted transporter exhibited significantly improved homogeneity and stability. Cryo-EM grid quality and particle distribution were markedly enhanced, enabling high-resolution structure determination and accelerating the client's structure-based drug discovery program.
Discuss Your Project with Our Experts
References:
Fill out this form and one of our experts will respond to you within one business day.