Membrane proteins are central to cellular signaling, transport, and energy transduction and represent the largest class of drug targets in modern therapeutics. However, their intrinsic hydrophobicity, structural complexity, and typically low expression levels make recombinant membrane protein production exceptionally challenging. Profacgen offers an integrated, end-to-end solution for membrane protein expression, isolation, stabilization, and purification, combining optimized expression platforms with advanced solubilization and reconstitution technologies. Our services enable reliable production of functional membrane proteins suitable for structural biology, biophysics, antibody discovery, and drug screening.
Membrane proteins play essential roles in fundamental biological processes and disease mechanisms, accounting for over 60% of current drug targets, including GPCRs, ion channels, transporters, and receptors. Despite their importance, recombinant production of membrane proteins remains one of the most technically demanding areas of protein science.
Two primary challenges dominate membrane protein production:
To overcome these barriers, Profacgen focuses on systematic optimization of both expression and purification, integrating advanced host systems, detergent screening strategies, and lipid-based stabilization technologies to preserve native structure and activity.
Profacgen provides a modular yet fully integrated membrane protein production pipeline, allowing clients to select individual services or a complete turnkey solution.
| Service Module | Service Description | Key Advantages | Typical Applications |
|---|---|---|---|
Recombinant Membrane Protein Production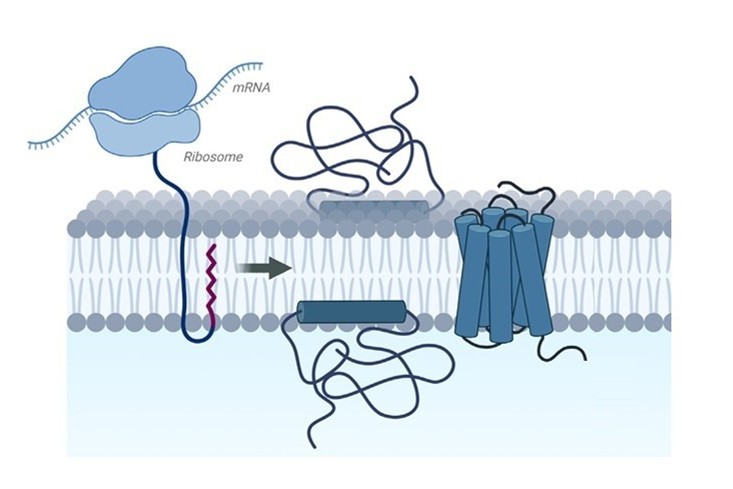 |
Customized expression of membrane proteins (e.g., GPCRs, ion channels, transporters, viral envelope proteins) using insect, mammalian, or cell-free expression systems. Construct design and expression strategies are optimized based on protein topology and functional requirements. | Flexible host selection; supports complex and multi-pass membrane proteins; preserves native folding and activity | Functional assays, structural biology, drug screening, antibody generation |
Enveloped VLPs Technology for Membrane Protein Production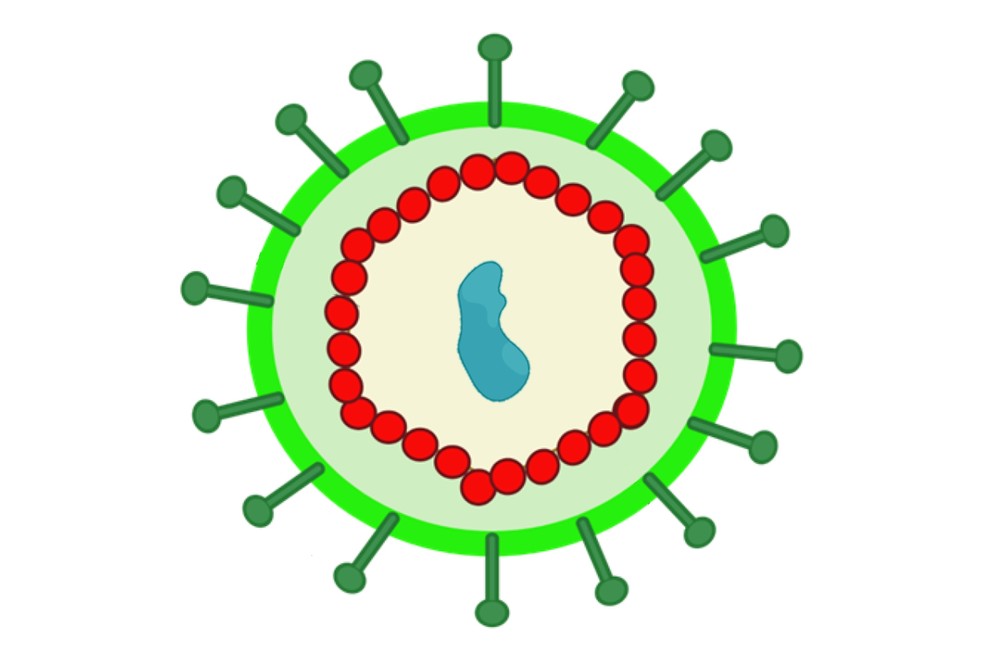 |
Incorporation of target membrane proteins into lipid bilayers during enveloped virus-like particle (VLP) assembly, enabling native membrane topology and oligomeric structure. | Native-like membrane environment; improved stability and antigenicity; enhanced functional relevance | Vaccine development, immunogenicity studies, receptor–ligand interaction analysis |
Membrane Protein Isolation and Purification |
Tailored extraction and purification workflows using affinity, ion-exchange, and size-exclusion chromatography under carefully controlled conditions to maintain protein integrity. | High purity and homogeneity; minimized aggregation; scalable purification strategies | Biochemical characterization, biophysical studies, downstream formulation |
Detergent Screening for Protein Purification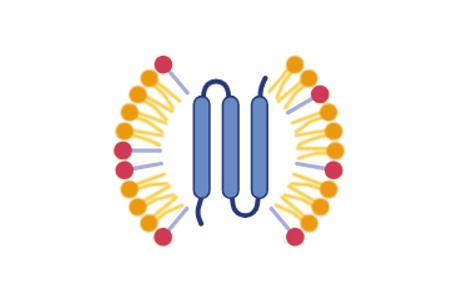 |
Systematic screening of non-ionic, zwitterionic, and mild ionic detergents to identify optimal solubilization conditions for each target membrane protein. | Protein-specific optimization; improved solubility and stability; reduced trial-and-error | Structural studies, cryo-EM, functional reconstitution |
Nanodisc-Based Protein Purification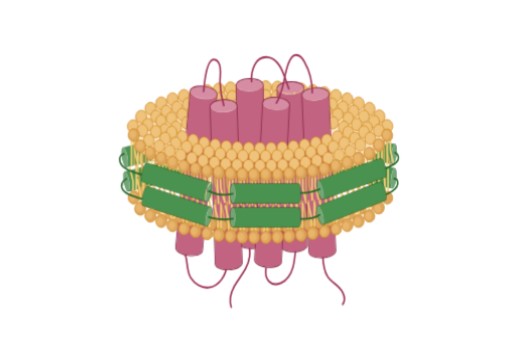 |
Reconstitution of purified membrane proteins into lipid nanodiscs composed of defined phospholipids and membrane scaffold proteins, closely mimicking native membranes. | Superior stability vs. detergents; native-like conformation; ideal for single-particle analysis | Cryo-EM, ligand binding studies, drug discovery, single-molecule assays |
Background
A pharmaceutical company required milligram-scale production of a human G protein–coupled receptor (GPCR) for cryo-EM structure determination and ligand-binding analysis. Previous expression attempts in bacterial systems resulted in severe aggregation, low yield, and loss of receptor stability, making the material unsuitable for high-resolution structural studies.
Our Solution
Profacgen implemented a baculovirus–insect cell expression strategy using optimized constructs and infection conditions. Following expression, a systematic detergent screening was performed to identify conditions that preserved receptor integrity. The GPCR was subsequently reconstituted into nanodiscs to further enhance stability and mimic the native membrane environment.
Final Results:
The purified GPCR displayed high homogeneity, excellent stability, and retained ligand-binding activity. The material enabled successful cryo-EM data collection and high-resolution structure determination.
Background
An academic research group sought functional expression of a 12-pass transmembrane transporter protein for substrate transport and inhibitor screening assays. The protein showed poor expression levels and rapid degradation in microbial systems, and initial purification attempts resulted in low recovery and instability.
Our Solution
Profacgen selected an insect cell expression platform to support proper membrane insertion and folding. Expression parameters were optimized to improve yield, followed by a comprehensive detergent screening program to identify solubilization conditions that maintained protein stability and activity throughout purification.
Final Results:
The transporter protein was obtained at high purity with preserved functional activity. The material was successfully applied in biochemical transport assays and biophysical characterization, enabling downstream mechanistic and screening studies.
Background
A biotechnology company required native-like presentation of a viral envelope membrane protein for antibody screening and immunogenicity evaluation. Conventional soluble expression approaches failed to reproduce correct protein conformation and membrane topology.
Our Solution
Profacgen employed enveloped virus-like particle (VLP) technology to incorporate the target membrane protein into a lipid bilayer during particle assembly. Expression conditions were optimized to ensure correct protein orientation, oligomerization, and stable VLP formation.
Final Results:
The resulting VLPs displayed the viral envelope protein in a native-like membrane context and demonstrated strong, specific antibody binding. This platform significantly accelerated antibody discovery and downstream immunological studies.
Consult Our Experts on Your Project
→ Learn more about our membrane protein platform: A New Platform for Membrane Proteins Expression
Contact us to discuss your membrane protein expression and purification project. Our technical and customer service teams are available to review your project requirements and design a customized solution that supports your research goals.
References:
Fill out this form and one of our experts will respond to you within one business day.