Ubiquitylation occurs with exquisite spatial, temporal and substrate specificity and has been linked to virtually every cellular process, including internalization, lysosomal targeting, modulation of protein interactions, alteration of subcellular distribution, regulation of transcription, DNA repair and propagation of transmembrane signaling. In addition, E3 ligases are implicated in a number of pathophysiological conditions, which makes them attractive therapeutic targets.
More than 600 E3 ligases have been observed and only a few of E3 ligase have been used in protein degrader designs. With our phage screening platform and small molecule library, we can provide a novel E3 ligases discovery service and used in protein degrader design.
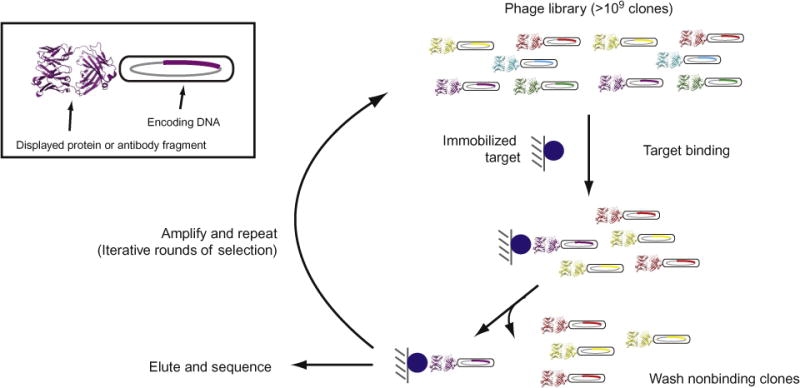
Figure 1. Phage display selection workflow
Phage display is a powerful selection technique that allows for the rapid selection of proteins with desired properties including increased affinity, specificity, stability etc. Here we can immobilize our E3 ligand and screening a higher affinity E3 ligase.
As an experienced leader in biology and pharmaceutical, Profacgen offers novel E3 ligases discovery services contributing to your research. Please contact us for more information.
1. Metzger MB, Hristova VA, Weissman AM. (2012). HECT and RING finger families of E3 ubiquitin ligases at a glance. Journal of Cell Science, 125(3), 531–537. doi:10.1242/jcs.091777
2. J.C. Frei and J.R. Lai. Protein and Antibody Engineering by Phage Display. Methods Enzymol. 2017; 580: 45–87. doi: 10.1016/bs.mie.2016.05.005
Fill out this form and one of our experts will respond to you within one business day.