Rabbit reticulocyte lysate (RRL) cell-free protein expression is a highly efficient eukaryotic in vitro translation system designed for rapid synthesis of complex proteins from RNA templates. Derived from highly specialized reticulocytes, which naturally synthesize hemoglobin, this system provides a robust and flexible platform to translate foreign RNA generated in vitro or introduced directly. Profacgen's optimized RRL platform, including coupled transcription–translation technology, enables streamlined single-tube reactions, significantly reducing time while maintaining high protein yield. The RRL system is ideal for applications requiring rapid functional protein synthesis, high-throughput screening, parallel expression, and studies of eukaryotic protein biogenesis, folding, and post-translational modifications.
Cell-free protein synthesis (CFPS) has been used for decades as a rapid, flexible, and cost-effective alternative to traditional cell-based protein production. CFPS systems provide direct access to the translational machinery of cells, allowing protein synthesis without the constraints of cell viability, growth, or long culturing periods. Among eukaryotic CFPS platforms, rabbit reticulocyte lysate is distinguished by its efficiency, speed, and compatibility with in vitro RNA templates.
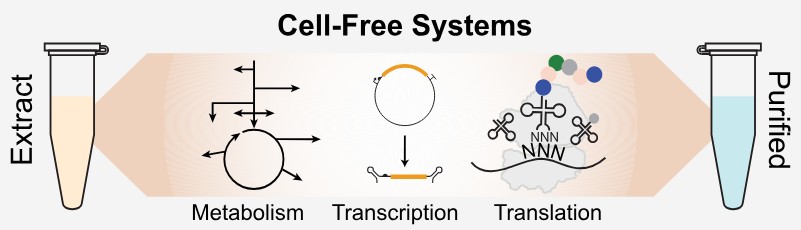 Figure 1. Cell-free gene expression. (Hunt et al., 2025)
Figure 1. Cell-free gene expression. (Hunt et al., 2025)
Reticulocytes are immature red blood cells highly specialized for hemoglobin production. The lysates derived from these cells retain ribosomes, tRNAs, translation factors, and other components necessary for efficient protein synthesis. Traditionally, RRL CFPS involves three separate reactions: in vitro transcription using SP6, T3, or T7 RNA polymerases, purification of the resulting RNA, and subsequent translation in the lysate. While effective, this multi-step process can be time-consuming and labor-intensive.
Profacgen has optimized this process by developing a coupled transcription–translation rabbit reticulocyte lysate system, which integrates transcription and translation into a single reaction tube. This approach streamlines the workflow, reduces handling steps, and allows for rapid synthesis of functional proteins directly from DNA templates, supporting both plasmid and PCR-generated constructs. The system is highly versatile, supports incorporation of labeled methionine for detection, and allows fine-tuning of reaction conditions such as Mg2+, K+, and DTT to maximize protein yield.
The RRL system is particularly suitable for the synthesis of proteins that are difficult to express in bacterial or eukaryotic cell lines, such as membrane proteins, regulatory factors, and proteins that require rapid functional testing. Its high protein synthesis rate, ability to express multiple proteins simultaneously, and compatibility with high-throughput workflows make it an ideal platform for both research and preclinical applications.
Profacgen offers a comprehensive suite of services using the rabbit reticulocyte lysate system, tailored to meet the specific needs of each protein and project.
Traditional RRL Cell-Free Translation
Single-Tube Coupled Transcription–Translation
Customized Reaction Optimization
Expression of Multiple Proteins Simultaneously
High-Throughput and Parallel Expression
Protein Purification and Technical Support
| Step | Details |
|---|---|
| Lysate Preparation | High-quality rabbit reticulocyte lysates are prepared from reticulocytes harvested from rabbits. Gentle lysis preserves ribosomes, tRNAs, translation factors, and other essential cellular machinery. |
| Reaction Assembly | Amino acids, energy substrates, RNA polymerase (SP6, T3, T7), and optional cofactors are added to the lysate to create the translation-ready system. For coupled transcription–translation reactions, DNA templates are transcribed in situ, producing mRNA directly in the reaction mixture. |
| Template Addition | Plasmid DNA or PCR-generated templates are introduced. In coupled transcription–translation reactions, DNA templates are transcribed in situ, producing mRNA directly in the reaction mixture. |
| Protein Synthesis | mRNA is translated by the translational machinery of the RRL system. Optional labeled amino acids can be incorporated for detection or functional studies. |
| Optimization and Recycling | Released mRNA can be recycled for multiple rounds of translation to increase yield. Reaction parameters can be fine-tuned with Mg2+, K+, or DTT to maximize protein production and stability. |
| Protein Purification | Target proteins are isolated from the reaction mixture. Depending on project requirements, purification strategies may include affinity tags, precipitation, or chromatography. |
Client Requirement:
A pharmaceutical research team required milligram quantities of a human cytokine for high-throughput functional screening. Conventional cell culture workflows demanded transfection, expansion, and harvest over several days, with yields fluctuating unpredictably between batches. These delays threatened timely progression of their drug discovery pipeline.
Our Solution:
We deployed a coupled transcription–translation rabbit reticulocyte lysate system, a eukaryotic cell-free platform supporting transcription and translation directly from plasmid DNA. A single-tube reaction eliminated transfection and culture steps entirely. Magnesium and potassium ion concentrations were systematically optimized to maximize soluble yield while preserving disulfide bond formation essential for cytokine activity.
Final Results:
Within 4–6 hours of template submission, the client received purified, bioactive cytokine at milligram scale. Cell-based proliferation assays confirmed full functional equivalence to mammalian-cell-derived reference material. The rapid turnaround enabled immediate integration into screening campaigns, compressing weeks of production time into a single day and allowing the client to meet critical program milestones without interruption.
Client Requirement:
A biotechnology company investigating signal transduction complexes required simultaneous production of four membrane-associated proteins for interaction mapping. Each construct possessed distinct hydrophobic characteristics and folding requirements; cell-based expression yielded only one soluble candidate after months of optimization, with the remaining three aggregating irreversibly.
Our Solution:
We implemented a multi-well coupled transcription–translation rabbit reticulocyte lysate workflow supporting parallel transcription and translation. Reaction parameters—including detergent supplementation, redox potential, and incubation temperature—were tailored individually to accommodate each protein's biochemical properties. The eukaryotic lysate provided native chaperones and translocation-competent microsomes where required.
Final Results:
All four targets were expressed successfully within a single 6-hour experiment. Co-immunoprecipitation confirmed complex formation and proper folding. By replacing sequential cell-based trials with parallel cell-free synthesis, the client reduced project timeline from months to weeks and eliminated the cost of repeated cloning and culture optimization, enabling rapid hypothesis testing.
Consult Our Experts on Your Project
Reference:
Fill out this form and one of our experts will respond to you within one business day.