Protein–protein interactions (PPIs) play essential roles in almost all cellular processes, including gene regulation, metabolic control, signal transduction, and cell communication.
Erroneous PPI is associated with a number of diseases (e.g., cancer, autoimmune diseases). Knowledge on the precise amino acid sequence that is responsible for the binding interaction is critical for the understanding of the binding mechanism and will largely facilitate targeted drug design.
Surface plasmon resonance (SPR) spectroscopy is a rapidly developing technique for the study of binding events involving proteins, which are the major molecular targets for validated drugs and for current drug discovery. This optical method measures the refractive index near a sensor surface and exploits total internal reflection of light at a surface-solution interface to produce an electromagnetic field or evanescent wave that is responsive to a binding event. SPR is label-free and capable of measuring real-time quantitative binding affinities and kinetics for protein interacting with ligand molecules using relatively small quantities of materials. At profacgen, our scientists have developed a high-throughput workflow for examining such interactions with an emphasis on identifying the binding site of a target protein. Based on the sequence of the target protein, overlapping peptides are synthesized as 15 or 20 mers which covers the full sequence of the protein. The peptide library is immobilized on the surface of a sensor chip and the analyte molecule (the interacting protein) is injected in the solution through the flow cell. By monitoring the change in the intensity of the reflected light (caused by the change in refractive index), peptides are screened against the analyte protein and those that can associate with the analyte are identified, which correspond to the regions of the target protein where binding occurs.
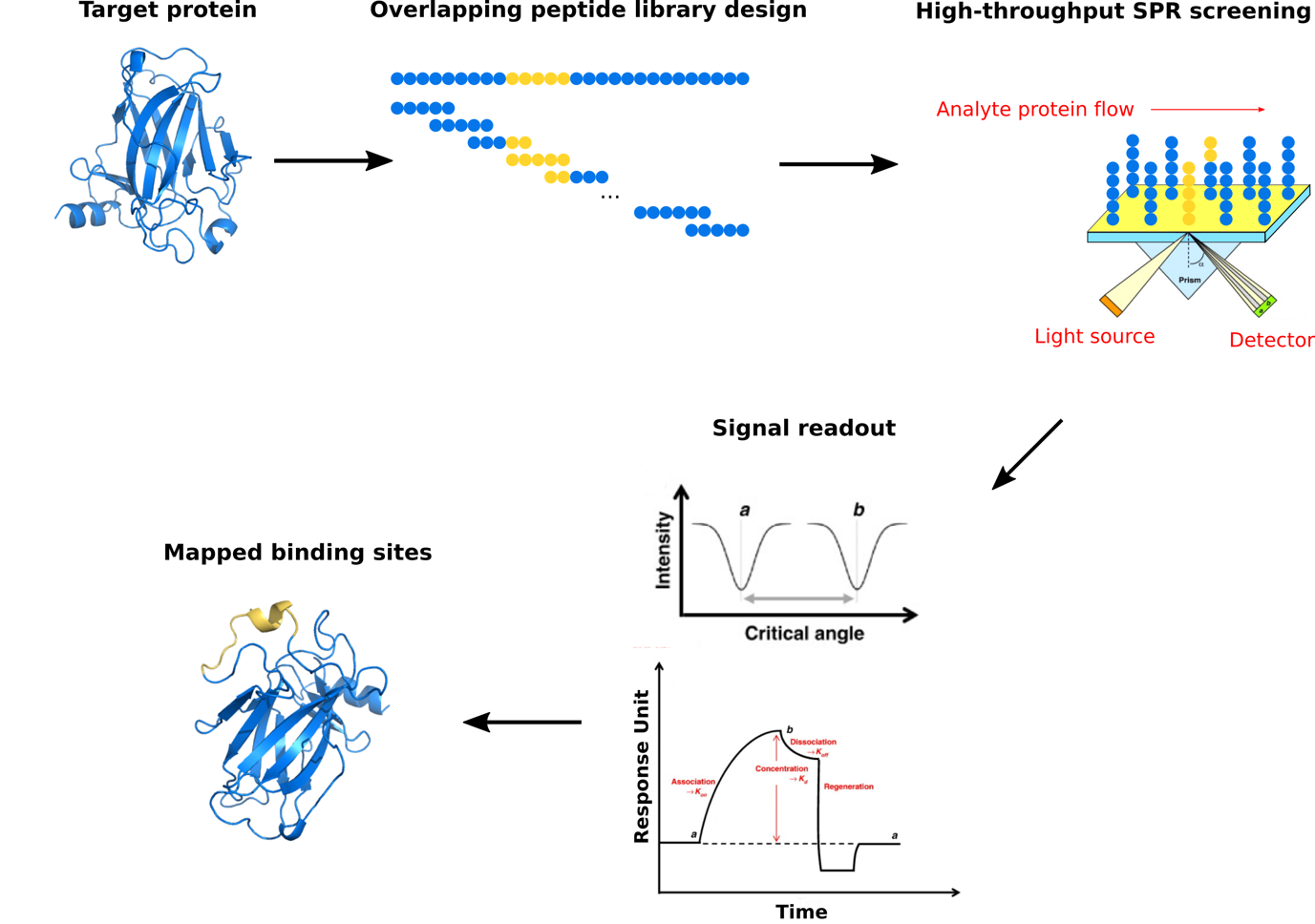
Profacgen employs SPR techniques to identify binding sites involved in protein-protein interactions. Our protein binding site mapping service is highly customizable, which suits our customers’ specific research goals. You only need to send us your target protein sequence and analyte protein sample(s) and we’ll perform all the necessary procedures from library preparation to data analysis. Our experienced scientists will work with you on the optimal screening strategy for your project and help design the peptide sequences.
High-throughput SPR technique is an ideal solution to map protein binding sites and characterize the molecular basis of the interaction in a fast and cost-effective manner. Please do not hesitate to contact us for more details about this service.
Fill out this form and one of our experts will respond to you within one business day.