Efficient protein production is a cornerstone of biopharmaceutical development, enzyme manufacturing, and industrial biocatalysis. A critical determinant of protein yield, folding, and secretion is the signal peptide—a short N-terminal sequence that directs nascent proteins to secretory pathways. While natural signal peptides have evolved for cellular fitness, they are rarely optimized for industrial-scale production. High-throughput engineering of signal peptides offers a transformative solution, combining combinatorial libraries, machine learning, and automated screening to systematically design sequences that maximize expression across diverse hosts, including Escherichia coli, yeast, and mammalian cells. This service accelerates protein development and unlocks higher yields with greater reliability. Profacgen offers high-throughput engineering of signal peptides to maximize protein expression efficiency and secretion performance.
Signal peptides are short amino acid sequences, typically 15–30 residues, located at the N-terminus of secreted or membrane-targeted proteins. They guide nascent proteins to the endoplasmic reticulum (ER) in eukaryotes or the periplasmic space in prokaryotes. Their primary functions include:
Despite their central role, natural signal peptides are rarely optimized for industrial expression. For example, the native Bacillus subtilis amylase signal peptide achieves only ~20% secretion efficiency in E. coli, whereas engineered variants can exceed 80%. Inefficient signal peptides can lead to misfolding, aggregation, retention in the cytosol, or heterogeneity—challenges that severely limit scale-up potential.
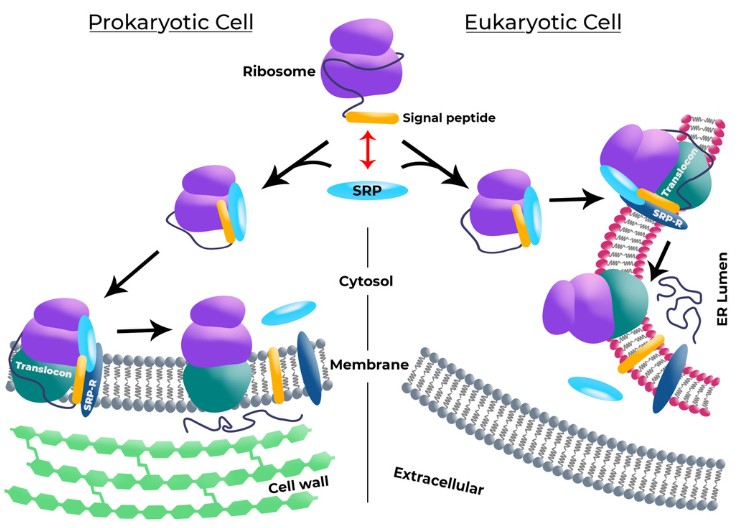 Figure 1. Signal peptides mechanism in prokaryotic cell and eukaryotic cell.
Figure 1. Signal peptides mechanism in prokaryotic cell and eukaryotic cell.
Traditional trial-and-error mutagenesis is laborious, limited in scope, and often fails to identify optimal sequences. High-throughput engineering overcomes these limitations, testing thousands of variants in parallel to rapidly identify the most effective signal peptides.
Profacgen provides a comprehensive high-throughput signal peptide engineering service that combines synthetic biology, computational design, and automated screening to optimize protein expression for any host system. Our platform is built to accelerate protein development while maximizing yield, solubility, and correct folding.
| Service Component | Key Features / Methods | Description / Benefit |
|---|---|---|
| Combinatorial Library Design |
|
Construct thousands of randomized or semi-rational signal peptide sequences. Enhance SRP binding, translocation efficiency, and engagement with the translocon. Incorporate host-specific protease recognition sequences. Broad sequence diversity allows identification of signal peptides that outperform natural sequences. |
| Machine Learning-Guided Design |
|
Analyze experimental data to identify sequence features correlated with high secretion. Optimize residues near cleavage sites and prevent premature folding. Match codon usage to host tRNA availability. Focus experimental efforts on the most promising candidates, reducing time and cost. |
| Automated High-Throughput Screening |
|
Quantitatively sort clones based on expression levels. Functional readouts measure secretion efficiency. Screen thousands of clones simultaneously in picoliter-scale reactors. Accelerates signal peptide discovery and ensures robust performance across multiple host systems. |
| Host-Specific Optimization | — | Tailor signal peptides for optimal performance in E. coli, yeast, insect, CHO, or other mammalian systems. Address host-specific secretion challenges to maximize protein yield. |
| Downstream Integration |
|
Integrate optimized signal peptides into your expression constructs. Support purification, detection, gene optimization, multimeric expression, and industrial-scale processes for seamless downstream application. |
Client Requirements:
A biopharmaceutical company was developing a therapeutic monoclonal antibody but experienced low secretion in CHO cells using the native signal peptide. Low yields limited functional assays, preclinical evaluation, and scale-up feasibility. The client required a solution that could reliably increase secretion without altering antibody functionality or glycosylation profiles.
Our Solution:
We designed a combinatorial library of signal peptides specifically optimized for CHO cells and applied machine learning-guided predictive modeling to focus on the most promising candidates. High-throughput screening using secreted reporter assays and ELISA quantified antibody secretion, and top-performing sequences were incorporated into the full-length antibody construct for validation.
Final Results:
Optimized signal peptides increased antibody secretion over threefold, maintaining correct folding and glycosylation. The enhanced yields supported high-throughput functional testing, accelerated preclinical timelines, and facilitated downstream process development for early-stage therapeutic evaluation.
Client Requirements:
An industrial biotech client aimed to produce a bacterial amylase at high yield for commercial enzyme applications. Expression using natural signal peptides resulted in misfolded protein accumulating in inclusion bodies, limiting recovery and driving up purification costs. The client needed a solution that maximized soluble secretion and scalability.
Our Solution:
We constructed a combinatorial library of signal peptides with optimized hydrophobic cores, N-terminal charges, and host-specific cleavage motifs. High-throughput microfluidic screening and secreted enzyme activity assays rapidly identified high-performing variants. Selected peptides were fused to the amylase gene and expressed under scalable E. coli conditions.
Final Results:
The engineered signal peptides increased soluble secretion from ~20% to over 85%. Misfolded aggregates were minimized, simplifying purification and reducing downstream costs. The improved system enabled consistent enzyme production at pilot scale, supporting commercial manufacturing.
Consult Our Experts on Your Project
Fill out this form and one of our experts will respond to you within one business day.