Membrane proteins—including receptors, ion channels, transporters, and enzymes—are essential regulators of cellular function and account for up to two-thirds of all druggable targets. Their pivotal roles in signal transduction, transport, and enzymatic activity make them critical in drug discovery. However, recombinant membrane proteins are notoriously challenging to produce in sufficient quantity and quality due to issues such as toxicity, aggregation, misfolding, and low yield. Profacgen offers a comprehensive recombinant membrane protein production platform, combining bacterial, yeast, mammalian, insect, and cell-free systems to produce functional, biologically active membrane proteins for research and therapeutic development.
Membrane proteins—including receptors, ion channels, transporters, and enzymes—are critical for cellular signaling, transport, and metabolism, representing up to two-thirds of druggable targets. Despite their importance, producing recombinant membrane proteins remains challenging due to their hydrophobic transmembrane regions, low endogenous abundance, and tendency to misfold or aggregate outside of lipid membranes. Overexpression can also be toxic to host cells, further limiting yields.
Many membrane proteins require complex folding and post-translational modifications, such as disulfide bonds and glycosylation, which often necessitate mammalian or eukaryotic expression systems. Purification is also difficult, as maintaining stability and activity typically involves detergents, lipid nanodiscs, or other membrane mimetics.
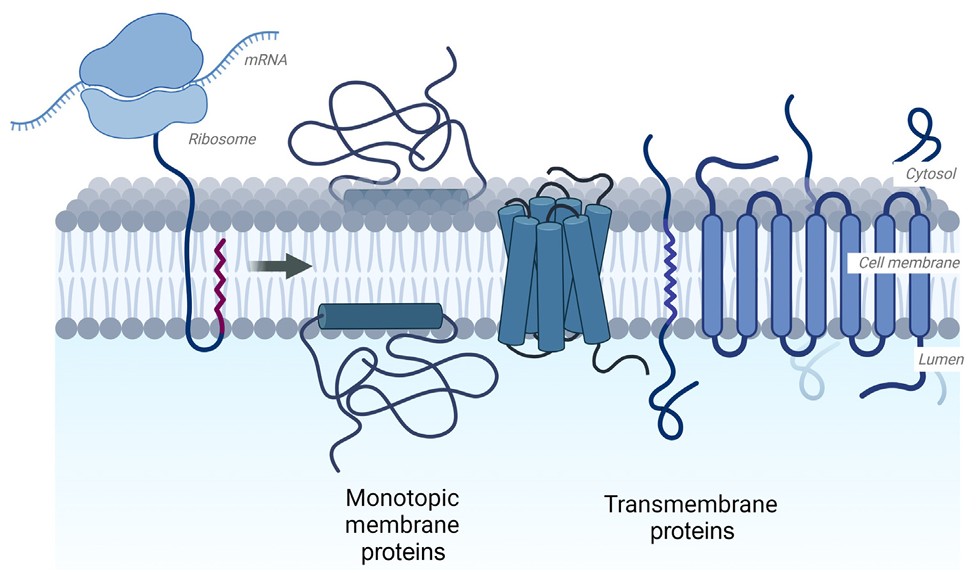 Figure 1. Membrane protein synthesis. (Manzer et al., 2023)
Figure 1. Membrane protein synthesis. (Manzer et al., 2023)
Advances in expression platforms, host engineering, vector design, and cell-free systems now allow for higher yields of functional, correctly folded membrane proteins. Profacgen integrates these strategies to deliver high-quality proteins suitable for drug discovery, structural biology, and therapeutic development.
Our recombinant membrane proteins are widely used for:
Profacgen provides a comprehensive suite of membrane protein production services, leveraging multiple expression platforms tailored to the protein type, complexity, and intended application. Our approach integrates advanced molecular biology, host cell engineering, and optimized purification strategies.



Baculovirus-Insect Expression System
Cell-Free Membrane Expression System
Tailored Optimization
Background
A pharmaceutical client required milligram quantities of a human G-protein-coupled receptor featuring seven transmembrane domains. Intended for high-throughput screening, the protein needed to retain native ligand-binding conformation. Endogenous expression was negligible, and previous efforts in standard systems resulted in severe aggregation and poor functional recovery.
Our Solution
We executed a parallel screening strategy using both yeast and mammalian expression platforms. Signal peptide engineering, gene dosage optimization, and inducible promoter selection were systematically evaluated. For material recovered from inclusion bodies, customized refolding protocols were developed to restore native architecture. Detergent screening during purification ensured membrane protein stabilization throughout the workflow.
Final Results
The campaign delivered approximately 15 mg/L of functional, properly folded GPCR. Radioligand binding assays confirmed expected affinity profiles, validating conformational integrity. The client successfully integrated this material into automated screening campaigns, directly accelerating hit identification. By providing high-quality functional protein without protracted cell line development, we shortened the client's drug discovery timeline by several months.
Background
A biotechnology company sought to determine the cryo-EM structure of a human voltage-gated ion channel to enable rational drug design. This multi-pass membrane protein was refractory to conventional overexpression; standard transfection produced predominantly misfolded aggregates unsuited for structural interrogation.
Our Solution
We deployed a baculovirus-mediated mammalian expression system, leveraging mammalian folding machinery to achieve native conformation. Transduction parameters—including multiplicity of infection, host cell line, and post-infection temperature shifts—were rigorously optimized. Media supplementation with small-molecule stabilizers and channel-specific ligands further enhanced protein integrity during production and extraction.
Final Results
The approach yielded 12 mg/L of monodisperse ion channel with electrophysiologically confirmed activity. Purified protein readily formed stable complexes suitable for single-particle analysis. The client subsequently obtained a high-resolution cryo-EM density map, revealing novel allosteric features. This enabled structure-guided medicinal chemistry efforts while avoiding the substantial investment required for stable cell line generation.
Background
An academic research team required a human multi-pass transporter for biochemical characterization. The protein exerted profound cytotoxicity when produced in living hosts—both bacterial and mammalian cultures arrested shortly after induction, precluding conventional fermentation.
Our Solution
We implemented a eukaryotic cell-free membrane protein system, eliminating host toxicity constraints entirely. The transporter was synthesized in supplemented lysates containing optimized detergent micelles, enabling co-translational insertion into lipid-like environments. Expression parameters, including lysate source, template concentration, and micelle composition, were rapidly screened within hours.
Final Results
Within 48 hours of project initiation, functional transporter was obtained at approximately 10 mg/L yield. Fluorescence-based transport assays confirmed correct membrane topology and substrate translocation activity. The client proceeded directly to kinetic characterization and mutagenesis studies without months of host engineering. This case demonstrates the unique power of cell-free platforms for accessing intractable, cytotoxic membrane targets.
Consult Our Experts on Your Project
Profacgen's recombinant membrane protein production services combine customized expression, large-scale production, flexible workflows, and high success rates to support research, drug discovery, and structural biology applications. Our expertise ensures rapid delivery of functional, high-quality membrane proteins, bridging the gap between challenging targets and successful therapeutic development. Please feel free to contact us for more information.
Reference:
Fill out this form and one of our experts will respond to you within one business day.