Animal-free protein expression systems provide a safe, scalable, and cost-effective alternative for recombinant protein production when the use of animal-derived materials must be avoided. Plant- and microalgae-based expression platforms significantly reduce the risk of contamination by animal-origin pathogens while offering strong advantages in scalability, storage stability, and downstream logistics. Profacgen delivers comprehensive animal-free protein expression solutions using advanced plant and microalgal systems, supporting applications ranging from research reagents and diagnostics to vaccines and biopharmaceutical development.
Traditional recombinant protein production often relies on microbial or mammalian cell systems, which may introduce concerns related to endotoxins, animal-origin contaminants, viral safety, or regulatory restrictions. Animal-free expression platforms address these challenges by eliminating direct contact with animal cells or animal-derived components.
Plant-based expression systems leverage the natural protein synthesis machinery of plants to produce recombinant proteins with proper folding and biological activity. Compared with microbial systems, plants support the expression of large and complex proteins. Compared with mammalian systems, they offer lower production costs, reduced biosafety risks, and simpler scale-up strategies. Importantly, plant-derived proteins are free from human and animal pathogens, making them particularly attractive for vaccine antigens, diagnostic reagents, and therapeutic proteins.
In addition to higher plants, microalgae have emerged as a next-generation animal-free expression platform. Microalgae combine the advantages of eukaryotic protein processing with rapid growth, controlled cultivation, and fully defined production conditions, offering excellent consistency and scalability.
Animal-free expression platforms provide ethically responsible, scalable, and contamination-free alternatives to traditional animal-derived systems. Profacgen offers multiple plant- and microalgae-based platforms to support diverse research and industrial protein production needs.

Lettuce Transient Expression System
The lettuce transient expression platform enables rapid production of recombinant proteins without the need to generate stable transgenic lines. Using Agrobacterium-mediated delivery or viral vectors, target genes are introduced into plant tissues for short-term, high-level expression.
This system is ideal for projects requiring fast turnaround, such as early-stage research, antigen screening, or feasibility studies. It supports the expression of soluble proteins, enzymes, and antigens, while maintaining an animal-free production process.
Nicotiana tabacum Expression System
Nicotiana tabacum is one of the most extensively studied plant expression hosts and is widely used for both transient and stable protein expression. Its large biomass, well-characterized genetics, and compatibility with Agrobacterium-mediated transformation make it a robust platform for producing recombinant proteins at scale.
Profacgen utilizes N. tabacum systems to express structurally complex proteins, vaccine antigens, and functional enzymes. Both leaf-based transient expression and stable transgenic approaches are available, allowing flexibility in production timelines and output scale.

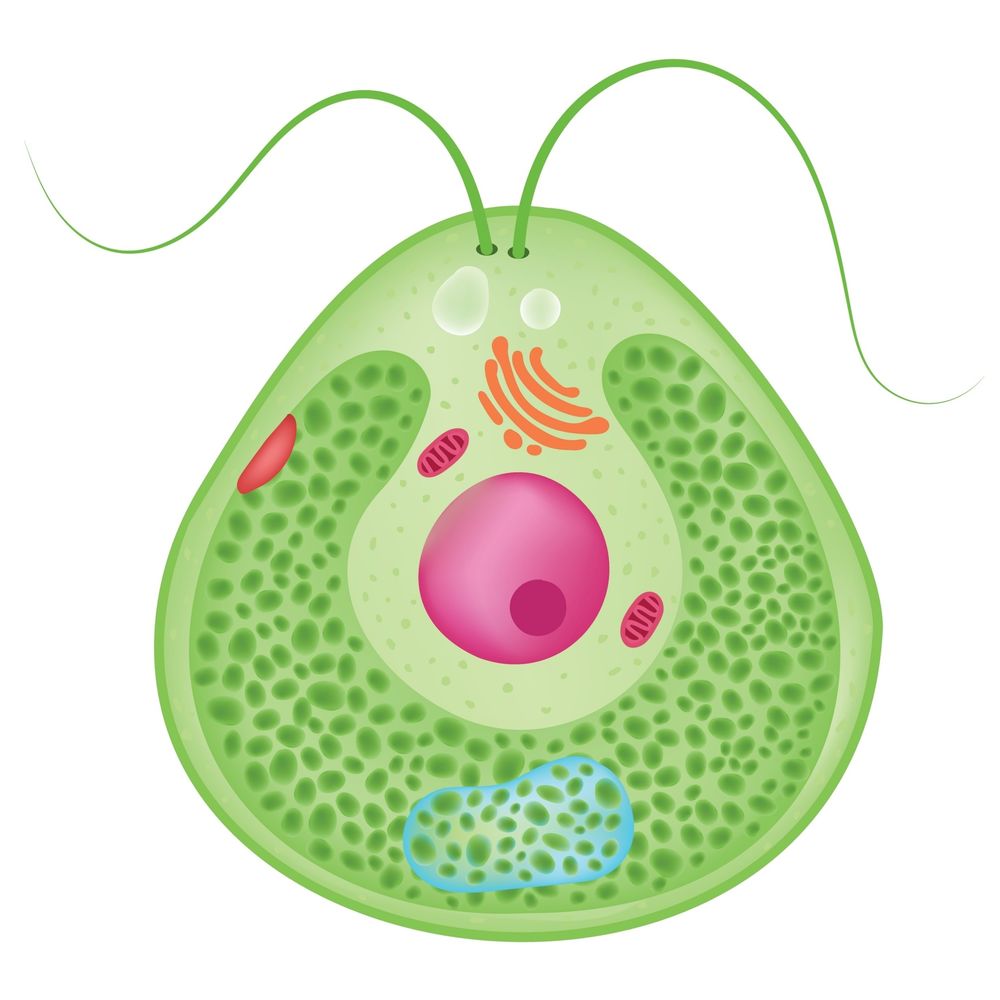
Microalgae Expression System
Microalgae expression systems offer a fully animal-free, contained, and environmentally sustainable production platform. These unicellular eukaryotes support proper protein folding and post-translational processing while enabling tightly controlled cultivation conditions.
Microalgae systems are particularly suitable for proteins requiring consistent quality, defined growth environments, and scalable bioreactor-based production. Applications include recombinant enzymes, antigens, and functional proteins for research, diagnostics, and industrial use.
While the above platforms provide flexible and scalable animal-free solutions, certain applications require enhanced protein stability, high production capacity, and cost-efficient long-term storage. For these needs, Profacgen offers a specialized and high-capacity rice endosperm–specific expression platform. This platform supports both short peptides (less than 100 amino acids) and large recombinant proteins (up to ~1000 amino acids).
Rice seeds provide a unique advantage for long-term storage and distribution, as recombinant proteins remain stable within the endosperm for extended periods without loss of bioactivity. Scale-up is achieved through standard agricultural cultivation, enabling economical large-scale production.
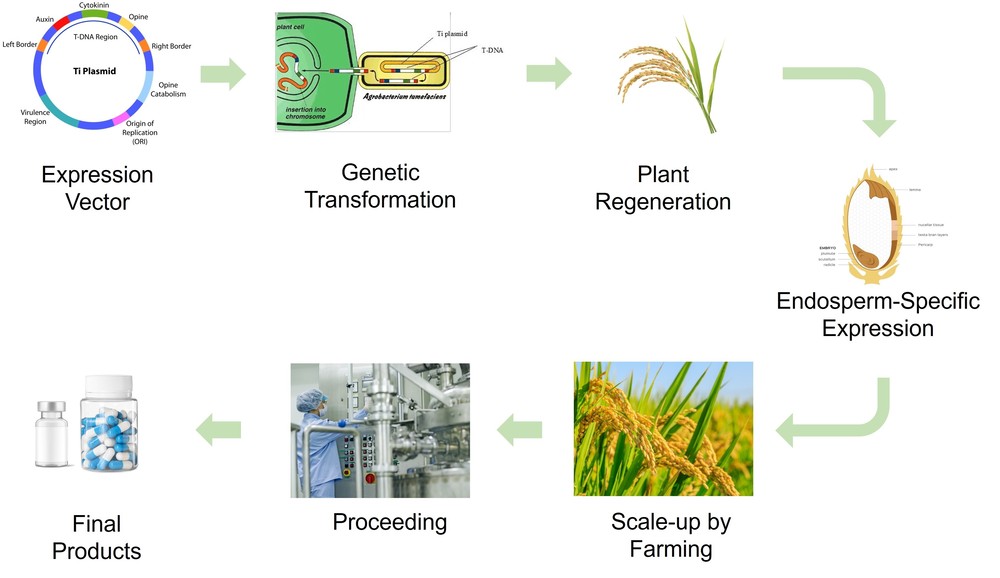 Figure 1. The rice endosperm-specific expression system.
Figure 1. The rice endosperm-specific expression system.
Key Features
| Phase | Service | Duration |
|---|---|---|
| Phase 1: Generation of Transgenic or Expression Hosts | Gene optimization and synthesis | 2 months |
| Vector construction and validation | 8 months | |
| Generation of transgenic plants or expression strains | 1~2 months | |
| Phase 2: Laboratory-Scale Evaluation | Screening of high-expression lines | 6 months |
| Genetic stability assessment | ||
| Protein expression and activity analysis | ||
| Development of purification protocols | ||
| Delivery of microgram-scale protein (90–95% purity) | ||
| Phase 3: Pilot-Scale Production | Production of milligram- to gram-scale protein | 6 months |
| Purity typically 90–95% | ||
| Biosafety assessment data for transgenic lines | ||
| Process development for scale-up and manufacturing |
Additional services include analytical method development, method validation, and downstream process optimization.
Background
A diagnostics company required an animal-free recombinant antigen for use in ELISA-based immunoassays. The protein needed to be free of animal-origin contaminants, demonstrate consistent batch-to-batch performance, and be scalable for long-term commercial supply. Speed was also a key consideration, as the antigen was required to support early assay development and validation.
Our Solution
Profacgen employed a Nicotiana tabacum transient expression system using Agrobacterium-mediated gene delivery. Expression constructs were optimized to enhance solubility and accumulation in plant tissue. Following expression, the antigen was purified using a streamlined protocol to ensure high purity and reproducibility.
Final Results
The antigen was produced at high yield and purity, showed strong immunoreactivity, and met all biosafety requirements. The material was successfully integrated into diagnostic assay development.
Background
A biotechnology company sought a cost-effective and animal-free platform for producing a recombinant therapeutic enzyme intended for long-term storage and global distribution. The client required gram-scale production, high protein stability, and minimal cold-chain dependency to support early-stage development and feasibility studies.
Our Solution
Profacgen developed a rice endosperm-specific expression system using Oryza sativa. Transgenic lines were generated and screened to identify high-expression, genetically stable homozygous plants. Purification protocols were optimized to recover active enzyme while maintaining product consistency.
Final Results
The enzyme was produced at gram scale with high purity and retained biological activity after extended storage in rice seeds. This approach significantly reduced production and storage costs while meeting the client's animal-free requirements.
Background
A vaccine research group required a fully animal-free expression platform for producing a recombinant antigen with high consistency and scalability. The protein was intended for immunogenicity testing and required controlled production conditions to ensure reproducible quality across batches.
Our Solution
Profacgen selected a microalgae expression system to enable eukaryotic protein processing in a contained, bioreactor-compatible environment. Gene constructs and cultivation parameters were optimized to maximize expression while maintaining protein integrity. Purification and analytical characterization were performed to confirm quality and consistency.
Final Results
The antigen was produced with excellent batch-to-batch reproducibility and preserved immunological properties. The material supported downstream immunogenicity studies and demonstrated the suitability of microalgae as a scalable animal-free platform.
Consult Our Experts on Your Project
Fill out this form and one of our experts will respond to you within one business day.