Multi-protein expression in insect cells, enabled by the Baculovirus Expression Vector System (BEVS), is a powerful and versatile approach for producing complex, functional protein assemblies with eukaryotic folding and post-translational modifications. By leveraging co-expression and co-infection strategies, multiple proteins can be simultaneously expressed and assembled into biologically active complexes within a controlled cellular environment.
At Profacgen, we provide a comprehensive multi-protein expression platform in insect cells, integrating advanced baculovirus engineering, flexible expression strategies, and optimized production workflows. Our services cover both single polycistronic baculovirus-based co-expression and multiple monocistronic baculovirus-based co-infection, as well as hybrid approaches. With extensive experience and a robust BEVS platform, we deliver high-quality protein complexes for structural biology, functional analysis, and biopharmaceutical development.
Multi-protein complexes play important roles in a wide range of cellular processes, including transcription, translation, RNA splicing, signaling cascades, protein trafficking, and degradation. These complexes function as coordinated molecular machines, enabling precise regulation of biological systems.
With the deepening of research, scientists have recognized that protein function is not solely determined by intrinsic physicochemical properties but is also heavily influenced by interactions with partner proteins and the overall architecture of protein assemblies. Consequently, increasing attention has been directed toward studying the structure and function of protein complexes, rather than individual proteins in isolation.
Protein complexes have demonstrated significant importance across multiple fields:
Reconstruction of multi-protein complexes through co-expression has become a widely used and effective strategy, allowing proteins to assemble in vivo into functional forms.
The Baculovirus Expression Vector System (BEVS) is a powerful platform for protein expression in insect cells such as Sf9 and Sf21.
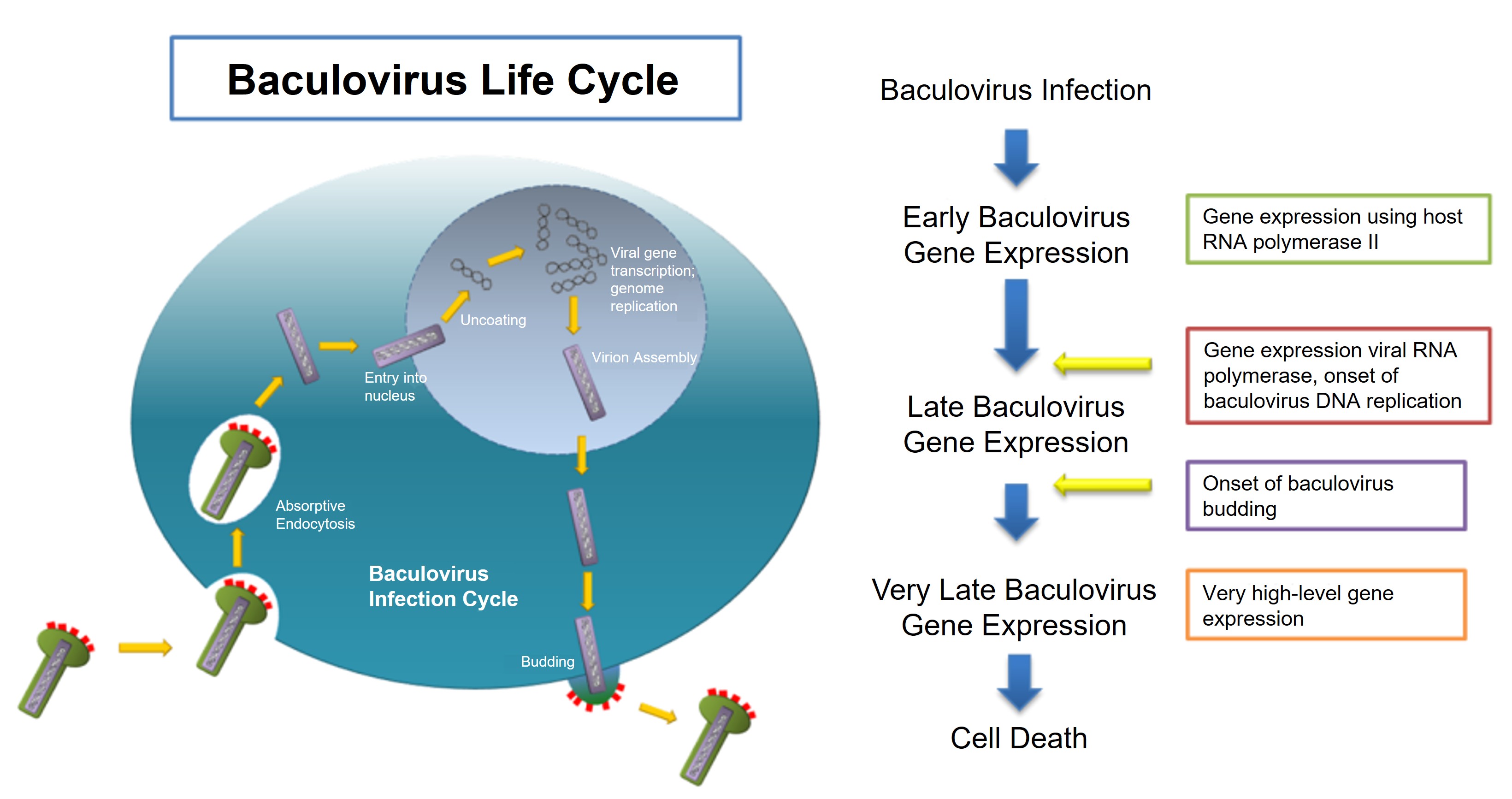 Figure 1. Baculovirus life cycle and gene expression.
Figure 1. Baculovirus life cycle and gene expression.
This system offers several key advantages:
Due to these advantages, BEVS has become a preferred platform for producing multi-protein complexes that require proper folding, assembly, and moderate post-translational modifications.
Profacgen provides a comprehensive, flexible, and advanced multi-protein expression platform in insect cells, covering both co-expression and co-infection strategies. Our services are designed to maximize protein yield, structural integrity, and biological functionality.

Profacgen offers multiple co-expression approaches to accommodate different experimental needs:
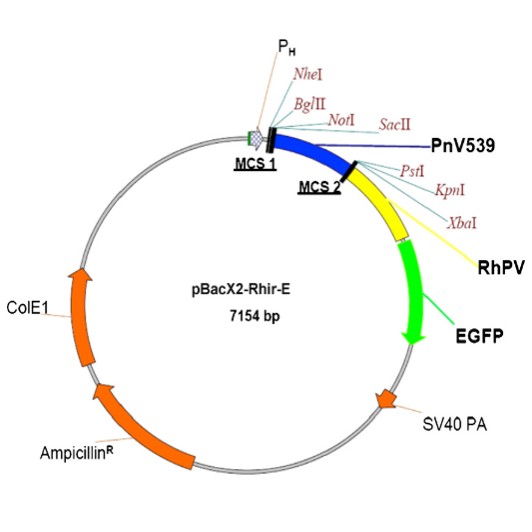
Single Polycistronic Baculovirus (Co-Expression)
In this approach, multiple genes are inserted into a single baculovirus vector, enabling coordinated expression of all target proteins.
Key Features
Advantages
Applications
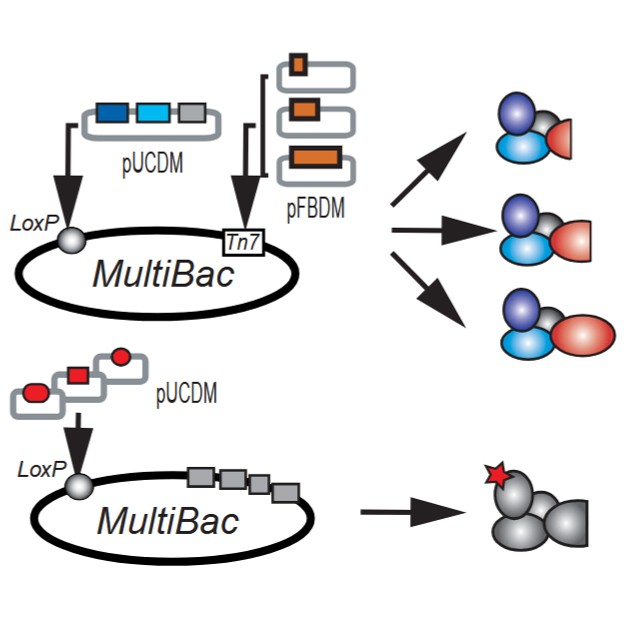
Multiple Monocistronic Baculoviruses (Co-Infection)
In this strategy, each gene is cloned into a separate baculovirus, and cells are co-infected with multiple viruses.
Key Features
Advantages
Applications
Hybrid Co-Expression and Co-Infection Strategies
Profacgen also offers combined approaches, integrating polycistronic and monocistronic systems to maximize flexibility and efficiency.
This hybrid strategy is particularly useful for:
BEVS-based multi-protein expression has been widely used in:
Client Requirements:
A research team required a multi-subunit enzyme complex for structural analysis by X-ray crystallography. Individual expression attempts yielded insoluble or improperly assembled protein, preventing complex formation and stalling their structural biology program.
Our Solution:
Profacgen designed a single polycistronic baculovirus for coordinated co-expression of all subunits in Sf9 insect cells, ensuring proper stoichiometry. Infection conditions including multiplicity of infection (MOI), cell density at infection, and harvest timing were systematically optimized to maximize yield of correctly assembled complex while minimizing proteolytic degradation.
Final Results:
Successfully produced a high-yield, soluble enzyme complex with correct assembly confirmed by size-exclusion chromatography and multi-angle light scattering. The purified complex diffracted well in crystallography trials, enabling structure determination and advancing the client's understanding of enzyme mechanism.
Client Requirements:
The client needed to analyze interactions among several signaling proteins where native stoichiometry was unknown, requiring flexibility to test multiple expression ratios during complex formation studies.
Our Solution:
We implemented a co-infection strategy using multiple monocistronic baculoviruses, each carrying a single gene. This approach allowed precise, independent control over expression levels by adjusting the MOI of each virus. Infection parameters were optimized to achieve desired subunit ratios while maintaining overall yield and cell health.
Final Results:
Achieved tunable expression enabling successful formation of multiple protein complexes with varying compositions. The flexibility allowed the client to map interaction networks and identify optimal complex stoichiometries for downstream functional studies, providing critical insights into pathway regulation.
Consult Our Experts on Your Project
Leveraging advanced baculovirus systems and optimized workflows, Profacgen delivers reliable and scalable solutions for multi-protein expression in insect cells. Our platform supports diverse co-expression and co-infection strategies to meet the demands of complex protein assembly and functional studies.
Contact us to start your insect cell expression project and benefit from customized solutions for efficient and high-quality protein complex production.
For applications focused on individual protein production rather than multi-subunit complexes, insect cell expression systems offer a flexible and scalable alternative. Our services support efficient expression with robust performance across a range of protein types.
References:
Fill out this form and one of our experts will respond to you within one business day.