Cell-free protein expression enables the synthesis of proteins in vitro using translation machinery extracted from cells. This versatile approach provides a rapid, efficient, and flexible alternative to conventional cellular protein production. By operating independently of living cells, it allows the high-yield production of toxic proteins, membrane proteins, and proteins prone to intracellular degradation. With the ability to manipulate reaction conditions directly, cell-free expression is ideal for high-throughput screening, protein engineering, and production of proteins with complex post-translational modifications (PTMs). Profacgen offers a complete, tailored solution for cell-free protein production. Our services can be customized to your specific needs, including tag selection, purity requirements, and more. With our expertise, you can expect high-quality proteins, rapid turnaround times, and competitive pricing, all designed to accelerate your research and development.
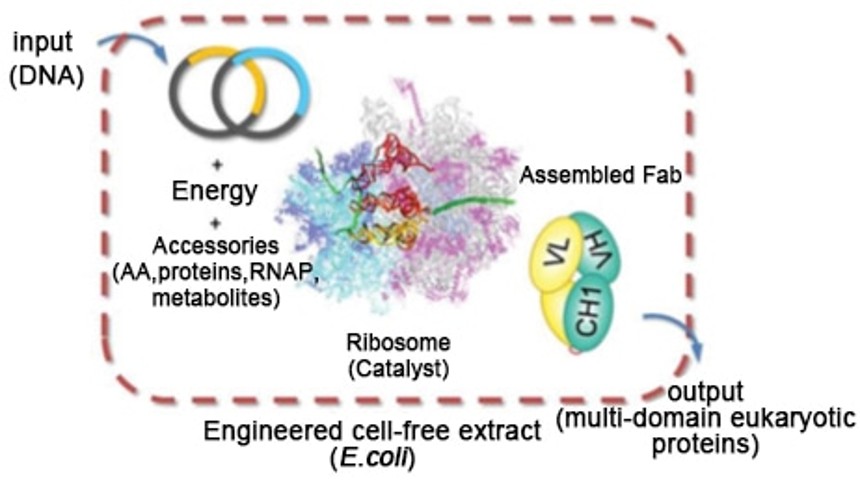
Cell-free protein expression systems utilize biological machinery from lysed cells to translate your gene of interest into functional proteins. Unlike conventional cell-based expression, these systems bypass cellular constraints, allowing precise control over the reaction environment. Typical reaction components include:
| Cell-Free System | Key Advantages | Typical Applications / Best Use Cases |
|---|---|---|
| E. coli Cell-Free Expression |
|
Bacterial proteins, enzyme variants, rapid prototyping, large-scale mutant or library screening |
| Rabbit Reticulocyte Lysate (RRL) |
|
Transcription factors, signaling proteins, functional assays requiring accurate translation |
| Wheat Germ Extract (WGE) |
|
Structural biology, protein–protein interaction studies, large eukaryotic protein expression |
| Insect Cell Lysate |
|
Membrane proteins, complex eukaryotic proteins, functional and biochemical assays |
| Tobacco Cell-Free Expression |
|
Plant proteins, plant metabolic enzymes, receptor studies |
| Human Cell Lysate |
|
Native human proteins, therapeutic research, functional studies requiring authentic PTMs |
Profacgen delivers an end-to-end solution for cell-free protein production, modeled after best practices from leading protein service providers and continuously optimized using in-house expertise.
Each platform can be customized based on protein size, complexity, PTM requirements, and downstream applications.
Background
A biotechnology company was conducting enzyme engineering and needed to rapidly express and evaluate more than 300 enzyme variants generated through site-directed mutagenesis. Their main challenges were the long cloning and cell culture timelines associated with conventional expression systems, as well as inconsistent expression levels that complicated activity comparison across variants.
Our Solution
Profacgen implemented an E. coli–based cell-free expression workflow optimized for parallel, high-throughput reactions. DNA templates were directly used without cell transformation, and reaction conditions were standardized to ensure reproducibility across all variants. Expression was completed within hours, enabling immediate downstream enzymatic assays.
Final Results
More than 95% of enzyme variants were successfully expressed at detectable levels. Overall screening time was reduced by over 70%, allowing the client to rapidly identify high-performance mutants and significantly accelerate their enzyme optimization program.
Background
An academic research group was studying a bacterial toxin protein that repeatedly caused host cell death when expressed in E. coli. Multiple attempts using different strains and induction conditions failed, resulting in either no expression or severe protein degradation, preventing further functional and structural characterization.
Our Solution
We applied a cell-free protein expression system that completely bypasses cellular viability constraints. Reaction conditions were carefully optimized to stabilize the toxic protein, including adjustment of energy components and reaction time. No living cells were involved, eliminating toxicity-related expression barriers.
Final Results
The target protein was successfully produced in soluble form with sufficient yield and stability. The protein was subsequently used for biochemical assays and functional validation, enabling the client to advance their research without the limitations imposed by cell-based expression systems.
Background
A pharmaceutical company required expression of a large, multi-domain human protein for functional studies. In mammalian cell systems, the protein showed low expression levels and extensive degradation, making it unsuitable for downstream assays and delaying project timelines.
Our Solution
Profacgen selected a wheat germ extract cell-free expression system known for its superior performance with large eukaryotic proteins. Reaction parameters were optimized to enhance translation efficiency and preserve protein integrity, while minimizing proteolytic degradation commonly seen in cellular systems.
Final Results
The full-length human protein was successfully expressed at high yield with intact functional domains. The resulting protein met the client's quality requirements and was directly applied to downstream functional and interaction studies, significantly shortening development timelines.
Consult Our Experts on Your Project
Fill out this form and one of our experts will respond to you within one business day.