Genotoxic Impurities Analysis
Genotoxic impurities (GTIs) refer to compounds that can cause DNA mutation, chromosome damage or DNA recombination at a very low concentration and potentially cause tumor development. Therefore, in recent years, many authoritative agencies such as FDA, EMEA, ICH, USP, CFDA have issued relevant guidelines, clearly stipulated the limits of genotoxic impurities. GTIs are required to be analyzed and controlled during the production of APIs and preparations. The control and detection of genotoxic impurities has attracted more and more attention of pharmaceutical companies, which play an important role in the process of drug development for the purpose of meeting the requirements of drug registration and declaration.
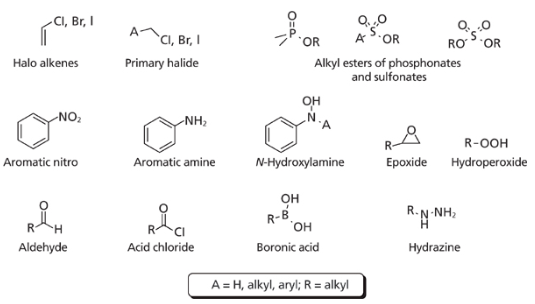
Figure 1. Structures of commonly observed GTIs.
Profacgen will perform your experiments based on the requirements of ICH. The determination of mutagenicity is mainly based on the results of the bacterial mutation test (Ames). Carcinogenicity is determined by the results of animal carcinogenicity tests and human carcinogenicity related evidences. In addition, other non-mutagenic genotoxic substances usually have a threshold mechanism. Such impurities usually do not pose a carcinogenic risk to humans when they exist at a certain level.
Services provided:
Provide development and verification of genotoxic impurity analysis methods.Evaluate genotoxic impurities that may be generated in the synthetic process route.
Use (Q) SAR prediction software to predict genotoxicity.
Evaluate genotoxicity by bacterial mutation experiment (Ames test).
Calculate the limits of genotoxic impurities in drugs according to the rules in ICH M7.
Evaluate genotoxic impurities in active pharmaceutical ingredients (APIs) based on ICH M7 guidelines and provide scientific control strategies.
Provide integrated methods to meet our customer needs.
We can provide various of methods for the analysis of GTIs. Two of the most commonly used techniques are GC and HPLC. In addition, we can also provide other techniques: GC-MS/MS, HS-GC-MS, LCMS, ICPMS, and IC.
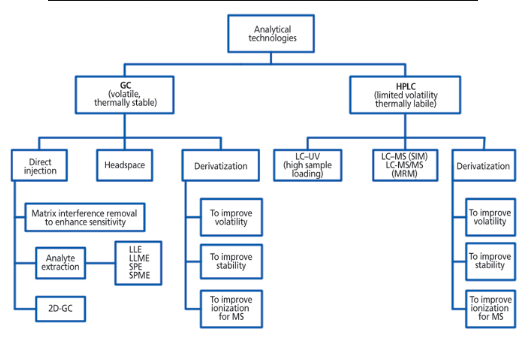
Figure 2. analytical method development strategy of GC and HPLC.
With the vast base of isolation techniques and characterization technologies in our GLP / GMP compliant protein analysis laboratories, underpinned by over 20 years of experience in supporting biologic drug development and bioprocessing, we offer a full spectrum of ready-to-use analytical methods for the detection and quantification of genotoxic impurities. Profacgen can ensure that you meet all regulatory expectations and specifications for your drug substance and drug product.
Reference:
1. Kumar. A., Zhang. K. and Wigman. L. (2015). Analytical technologies for genotoxic impurities in pharmaceutical compounds. LCGC North America. 33 (5), pp.344-359.