Biomembrane Force Probe (BFP)
The Biomembrane Force Probe (BFP) is a sensitive technique that allows the quantification of single molecular bonds. It is a versatile tool that can be used in a wide range of forces (0.1 pN to 1 nN) and loading rates (1–106 pN/s)[1]. BFP consists of a red blood cell sucked up by a capillary tube and a glass bead stuck in front of the red blood cell. Ligand molecules or antibodies corresponding to platelet surface receptor proteins are attached to the glass beads, which can cause adhesion with platelets.
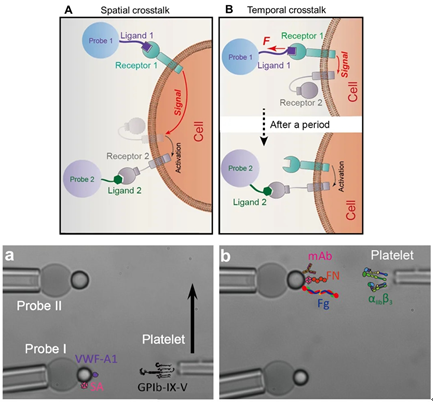
BFP is divided into traditional biofilm dynamometer and dual-load biofilm dynamometer. Compared with the traditional biofilm dynamometer with only one set of force measurement devices, the dual-loaded biofilm dynamometer has two sets of force measurement devices: Probe I and Probe II[2,3], so you can use Probe I to mechanically stimulate platelets and monitoring the activation state of platelets with Probe II. Using BFP, you can know the adhesion between platelets and glass beads by observing whether the red blood cells are pulled and lengthened, as well as the length and length of the red blood cells, and measure the receptor and ligand / antibody molecules Affinity and binding stability under different tensile forces.
Advantages
- Enables the investigation of the cooperative binding of multiple receptor–ligand species located in the proximal or separate regions of the same cell surface using mixture presentation or separate presentation of multiple ligands.
- Three single applications can be illustrated using four dual receptor biological systems with biological interests in two different fields: immunology and hemostasis/thrombosis.
- Can stimulate a single cell with soluble agonists without the influence of paracrine signaling from neighboring cells, reducing impact on biological macromolecular interactions and functions.
- Single-molecule level detection, high sensitivity, high precision, high dynamic range (dissociation constant Koff can reach 10-3 s-1, affinity dynamic detection range is 10 mM -1 nM, mechanical resolution ~ 1 pN)
Profacgen can provide single-molecule protein in situ dynamic detection technology to detect and analyze the dynamic interaction characteristics of in situ detection of cell membrane proteins, transmembrane proteins and their interacting molecules on the cell surface to obtain high-precision interaction affinity and dissociation constant and force parameters. It has important application value in the development of biological innovative drugs, and development and evaluation of biosimilar drugs.
References:
[1] Gourier, C., Jegou, A., Husson, J. et al. A Nanospring Named Erythrocyte. The Biomembrane Force Probe. Cel. Mol. Bioeng.1, 263 (2008). https://doi.org/10.1007/s12195-008-0030-x
[2] Ju, L., Chen, Y., Li, K. et al. Dual Biomembrane Force Probe enables single-cell mechanical analysis of signal crosstalk between multiple molecular species. Sci Rep 7, 14185 (2017). https://doi.org/10.1038/s41598-017-13793-3
[3] Chen, Y., Ju, L.A., Zhou, F. et al. An integrin αIIbβ3intermediate affinity state mediates biomechanical platelet aggregation. Nat. Mater. 18, 760–769 (2019). https://doi.org/10.1038/s41563-019-0323-6