The N- and C-terminal sequences of a protein define its translational boundaries, processing fidelity, and functional integrity. Profacgen's N-Terminal and C-Terminal Sequencing services integrate classical Edman degradation with advanced LC-MS/MS and top-down mass spectrometry platforms to deliver unambiguous terminal sequence confirmation. Whether verifying signal peptide cleavage, detecting post-translational modifications such as N-terminal acetylation or C-terminal amidation, or resolving truncation variants, our dual-platform approach ensures comprehensive coverage of both termini with high sensitivity and regulatory-grade documentation.
Accurate determination of protein termini is indispensable throughout biopharmaceutical development, from clone selection to lot-release testing. The N-terminus governs protein half-life, subcellular targeting, and immunogenicity, while the C-terminus frequently mediates protein–protein interactions and structural stability. Regulatory agencies expect terminal sequence verification as part of the identity and purity specification for recombinant therapeutics.
Profacgen addresses these requirements through a complementary dual-platform strategy. Edman degradation provides direct, database-independent N-terminal sequencing with unambiguous discrimination of isobaric residues such as leucine and isoleucine. For blocked or modified termini, our LC-MS/MS workflows—encompassing N-terminal peptide enrichment, chemical labeling, and high-resolution tandem mass spectrometry—recover sequence information that Edman chemistry cannot access. C-terminal sequencing is performed via carboxypeptidase time-course digestion combined with LC-MS/MS or MALDI-TOF analysis, enabling stepwise release and identification of C-terminal residues.
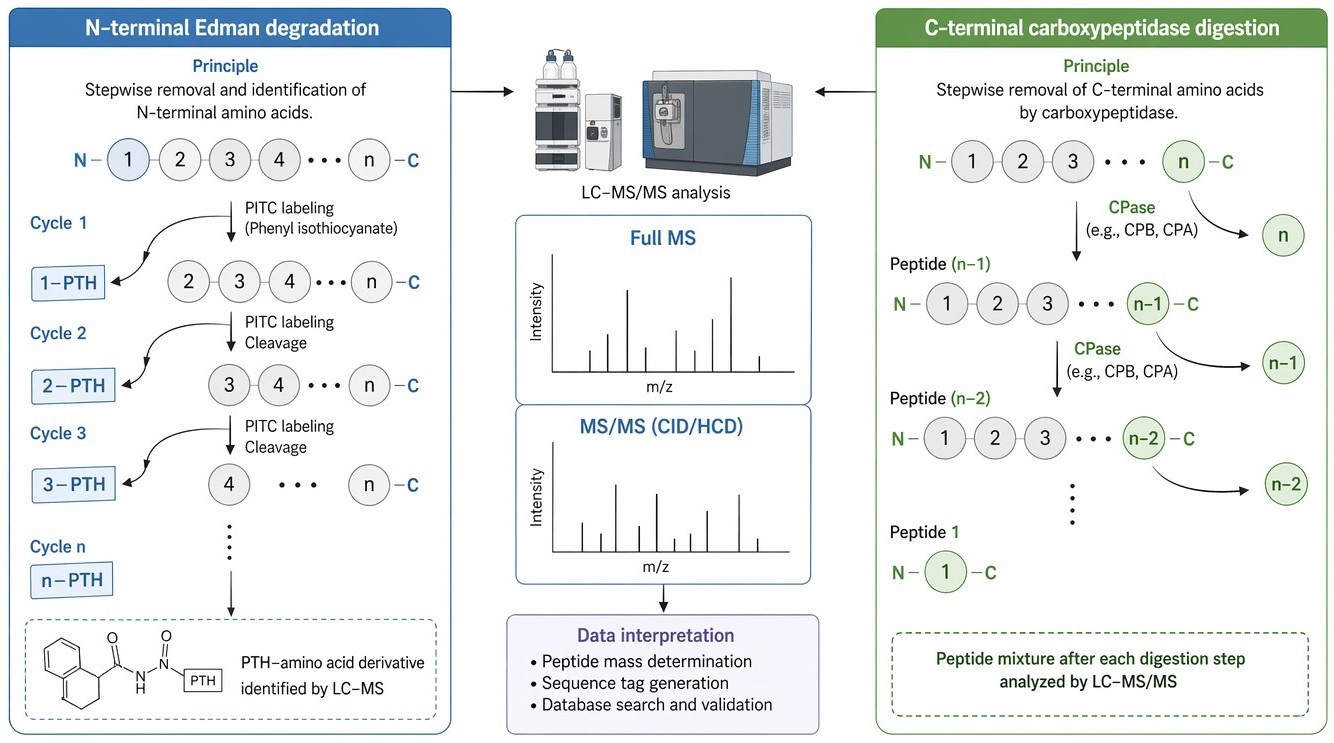 Figure 1. Schematic overview of N-terminal Edman degradation and C-terminal carboxypeptidase digestion coupled with mass spectrometric detection.
Figure 1. Schematic overview of N-terminal Edman degradation and C-terminal carboxypeptidase digestion coupled with mass spectrometric detection.
Profacgen provides comprehensive terminal sequencing solutions tailored to research, diagnostic, and biopharmaceutical quality control applications. Our offerings include:
| Service Component | Description |
|---|---|
| N-Terminal Sequencing by Edman Degradation |
|
| N-Terminal Sequencing by LC-MS/MS |
|
| C-Terminal Sequencing |
|
| Top-Down Intact Protein Terminal Mapping |
|
| Integrated Reporting & Regulatory Support |
|

Background:
A biopharmaceutical client observed heterogeneous charge profiles during ion-exchange chromatography of their Phase I monoclonal antibody. The anomaly threatened to delay IND filing because regulatory guidelines require demonstration of N-terminal consistency. Initial Edman attempts failed to yield a signal, suggesting a blocked N-terminus, but the exact modification remained unidentified.
Our Solution:
Profacgen deployed a combined analytical strategy. We first performed N-terminal peptide enrichment using dimethyl labeling after tryptic digestion, followed by LC-MS/MS on a Q-Exactive Orbitrap. Parallel top-down ETD-MS of the intact antibody provided complementary fragmentation. Both platforms consistently identified a mixture of N-terminal glutamine cyclization (pyroglutamylation) and partial N-terminal acetylation at the heavy chain, arising from upstream cell-culture conditions.
Final Results:
Quantitative analysis revealed 78 % pyroglutamylated, 15 % acetylated, and 7 % free N-terminus. The client used these data to optimize harvest time and media formulation, reducing pyroglutamylation to <5 % in subsequent batches. Profacgen's report, including raw MS files and validated methods, was incorporated directly into the regulatory CMC section, allowing the client to proceed with IND submission on schedule.
Background:
A contract development organization noted a 2 kDa mass discrepancy between the expressed recombinant cytokine and its theoretical molecular weight. SDS-PAGE and intact mass analysis confirmed the presence of a lower-molecular-weight species at approximately 15 % relative abundance. The client needed to determine whether the variant represented a C-terminal truncation, a critical quality attribute for potency and stability.
Our Solution:
We subjected the protein to time-course digestion with carboxypeptidase Y, sampling aliquots at 0, 5, 15, 30, and 60 minutes. Each time point was analyzed by MALDI-TOF to monitor mass shifts. Simultaneously, the intact protein and digested fragments were analyzed by bottom-up LC-MS/MS using AspN to generate C-terminal peptides. The combined data allowed us to reconstruct the precise C-terminal sequence loss.
Final Results:
Mass spectrometric tracking identified progressive removal of C-terminal residues, pinpointing a truncation site at the penultimate arginine residue. The truncated variant lacked the terminal RR motif essential for heparin binding, explaining the observed potency loss in bioassay. The client revised their downstream purification protocol to remove the truncated species, restoring specific activity to target specifications and securing batch release.
Background:
A gene-therapy startup engineered a secreted fusion protein carrying an immunoglobulin Fc domain and a growth factor. During early development, Western blotting detected an unexpected band approximately 3 kDa larger than predicted. The team suspected incomplete signal peptide cleavage, which would compromise secretion efficiency and expose non-native N-terminal sequences with potential immunogenicity implications.
Our Solution:
Profacgen performed Edman degradation on the intact fusion protein blotted onto PVDF after SDS-PAGE separation. The first five cycles yielded a sequence perfectly matching the predicted mature N-terminus (Glu-Pro-Met-Val-Thr), confirming correct signal peptidase processing. To rule out minor populations of uncleaved precursor, we conducted N-terminal peptide enrichment LC-MS/MS, which detected the signal peptide-containing species at <1 % abundance—well below the critical threshold for product specification.
Final Results:
The dual-platform confirmation satisfied both internal quality gates and external investor due diligence. The client proceeded to stable cell line development with confidence that the expression construct produced the intended mature protein. Profacgen's rapid 10-day turnaround prevented a two-month delay that would have resulted from alternative sequencing arrangements.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.