Purity and size variant analysis is essential for ensuring the safety, efficacy, and consistency of biopharmaceutical products. Profacgen's Purity & Size Variant Analysis services employ orthogonal chromatographic, electrophoretic, and light-scattering platforms to quantify high-molecular-weight species (HMWS), low-molecular-weight species (LMWS), and process-related impurities with regulatory-grade precision. Whether performing routine lot-release testing, monitoring stability trends, or conducting forced-degradation studies to support formulation development, our comprehensive platform delivers the quantitative data and documentation required to meet ICH Q6B, FDA CMC, and USP compendial standards.
Biopharmaceutical purity is not defined by a single attribute but by a panel of complementary measurements that collectively ensure product quality. Size variants—particularly aggregates and fragments—are classified as critical quality attributes (CQAs) because they can directly impact immunogenicity, potency, pharmacokinetics, and shelf-life stability. Regulatory agencies including the FDA, EMA, and ICH explicitly require specification of molecular size variants as part of drug substance and drug product release testing under ICH Q6B guidelines.
High-molecular-weight species (HMWS) include dimers, trimers, and higher-order aggregates that may form during cell culture, purification, or storage. These species are of particular concern because protein aggregates can elicit unwanted immune responses and alter bioactivity. Low-molecular-weight species (LMWS) typically arise from proteolytic clipping, disulfide reduction, or hinge-region fragmentation, generating species such as Fc-Fab (~100 kDa) and free Fab (~47 kDa) that may lack effector function or exhibit altered clearance. Profacgen addresses these challenges through a multi-method orthogonal platform that combines size-exclusion chromatography (SEC-HPLC/UHPLC), capillary electrophoresis with sodium dodecyl sulfate (CE-SDS), and SEC coupled with multi-angle light scattering (SEC-MALS) to achieve comprehensive, quantitative purity profiling.
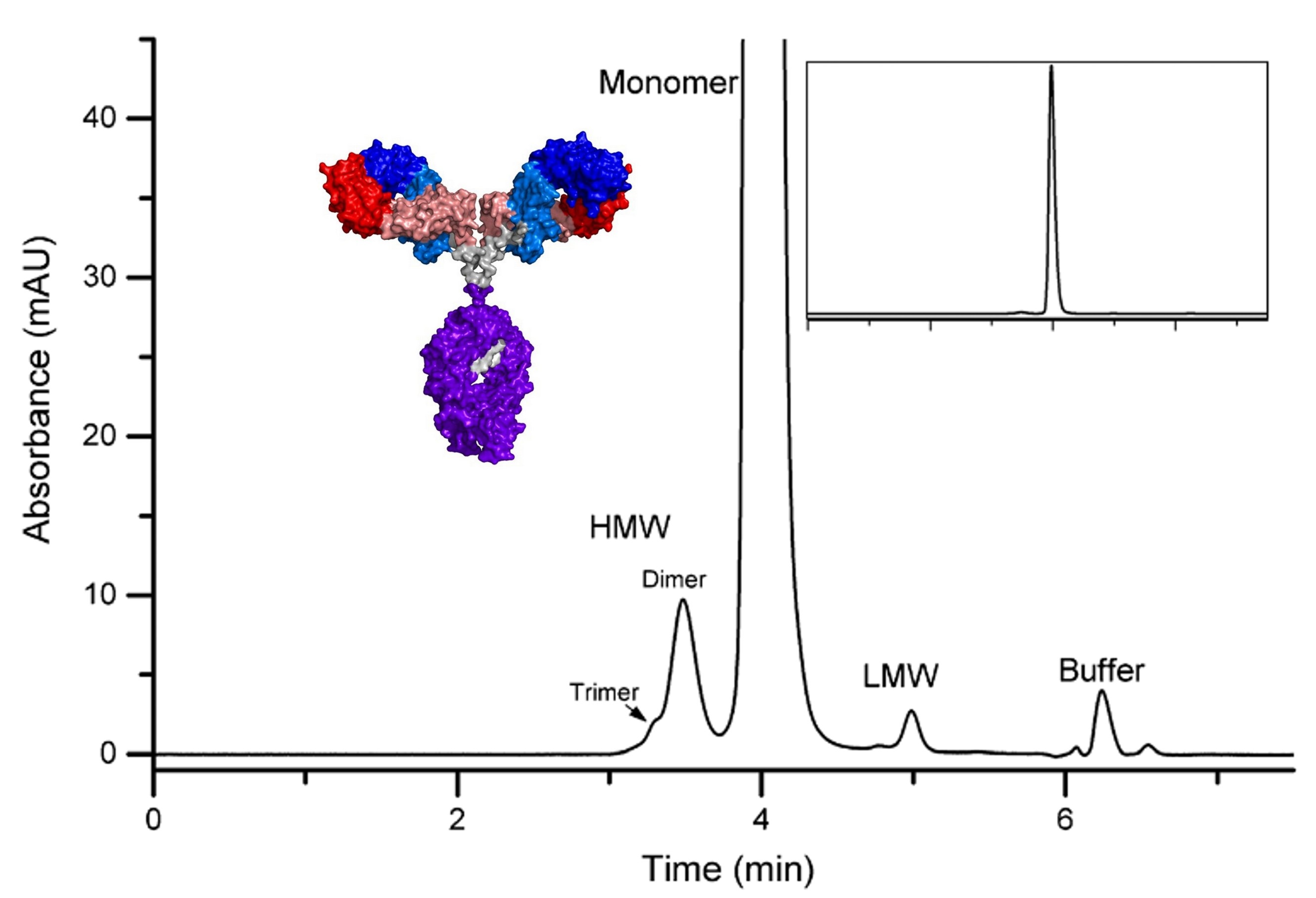 Figure 1. Representative SEC chromatogram for orthogonal NISTmAb size analysis. (Turner et al., 2018)
Figure 1. Representative SEC chromatogram for orthogonal NISTmAb size analysis. (Turner et al., 2018)
Profacgen provides end-to-end purity and size variant characterization tailored to research, development, and quality control applications. Our offerings include:
| Service Component | Description |
|---|---|
| Size-Exclusion Chromatography (SEC-HPLC / SEC-UHPLC) |
|
| Capillary Electrophoresis–SDS (CE-SDS) |
|
| SEC-MALS & Advanced Detection |
|
| Subvisible & Particulate Matter Analysis |
|
| Forced Degradation & Stability Studies |
|

Background:
A biosimilar developer needed to demonstrate analytical similarity between their trastuzumab candidate and multiple lots of the EU reference product. Size variant profiling was a critical component of the totality-of-evidence package because aggregate and fragment levels are tightly specified for the innovator product and directly linked to immunogenicity risk. The client required a platform SEC method capable of resolving HMWS and LMWS with baseline separation and quantitative precision.
Our Solution:
Profacgen implemented a validated platform SEC-HPLC method using a TSKgel G3000SWxl column with 0.2 M potassium chloride in 0.25 mM phosphate buffer (pH 7.0) at 0.5 mL/min. The method was qualified for specificity, repeatability, and linearity using an in-house IgG1 control standard. The biosimilar candidate and 21 EU reference lots were analyzed in triplicate. Peak integration employed a fixed baseline with perpendicular delimiter drop, and % HMWS, % monomer, and % LMWS were quantified from the total integrated peak area.
Final Results:
The platform method achieved baseline resolution between dimer and monomer (Rs > 2.6) for all samples. The biosimilar candidate exhibited 0.4 % HMWS, 99.2 % monomer, and 0.4 % LMWS—values that fell within the observed distribution of the 21 EU lots (HMWS: 0.3–0.7 %; monomer: 99.0–99.4 %; LMWS: 0.2–0.6 %). No new peaks were detected in the biosimilar chromatogram. The FDA accepted Profacgen's SEC comparability data as part of the analytical similarity assessment, and the client advanced to Phase III without additional size variant studies.
Background:
A clinical-stage biotech observed an upward trend in HMWS during accelerated stability testing of their Fc-fusion protein at 40 °C/75 % RH. Over six months, HMWS increased from 1.2 % to 4.8 %, approaching the release specification limit of 5.0 %. The client needed to determine the aggregation mechanism, identify whether the aggregates were covalent or noncovalent, and establish whether the trend would continue through the proposed 24-month shelf life.
Our Solution:
Profacgen performed a multi-method investigation. SEC-HPLC under native conditions quantified total HMWS. SEC-MALS was employed to determine the absolute molecular weight of the aggregate peak, revealing a species of approximately 300 kDa consistent with a noncovalent trimer rather than a covalent dimer. Non-reduced CE-SDS showed no increase in high-molecular-weight bands, confirming that the aggregates were held by noncovalent interactions rather than disulfide scrambling. Reduced CE-SDS detected a slight increase in a ~75 kDa clip species, suggesting that limited proteolysis preceded aggregation. DSC showed a 1.8 °C decrease in the fusion domain Tm in the stressed samples, indicating localized unfolding as the initiating event.
Final Results:
The combined data established a degradation pathway: localized thermal unfolding of the fusion domain exposed hydrophobic patches that drove reversible, noncovalent trimerization, with minor proteolytic clipping as a secondary event. The client used these findings to reformulate with 100 mM proline as a stabilizing excipient, which suppressed HMWS formation to <2.0 % at 24 months.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.