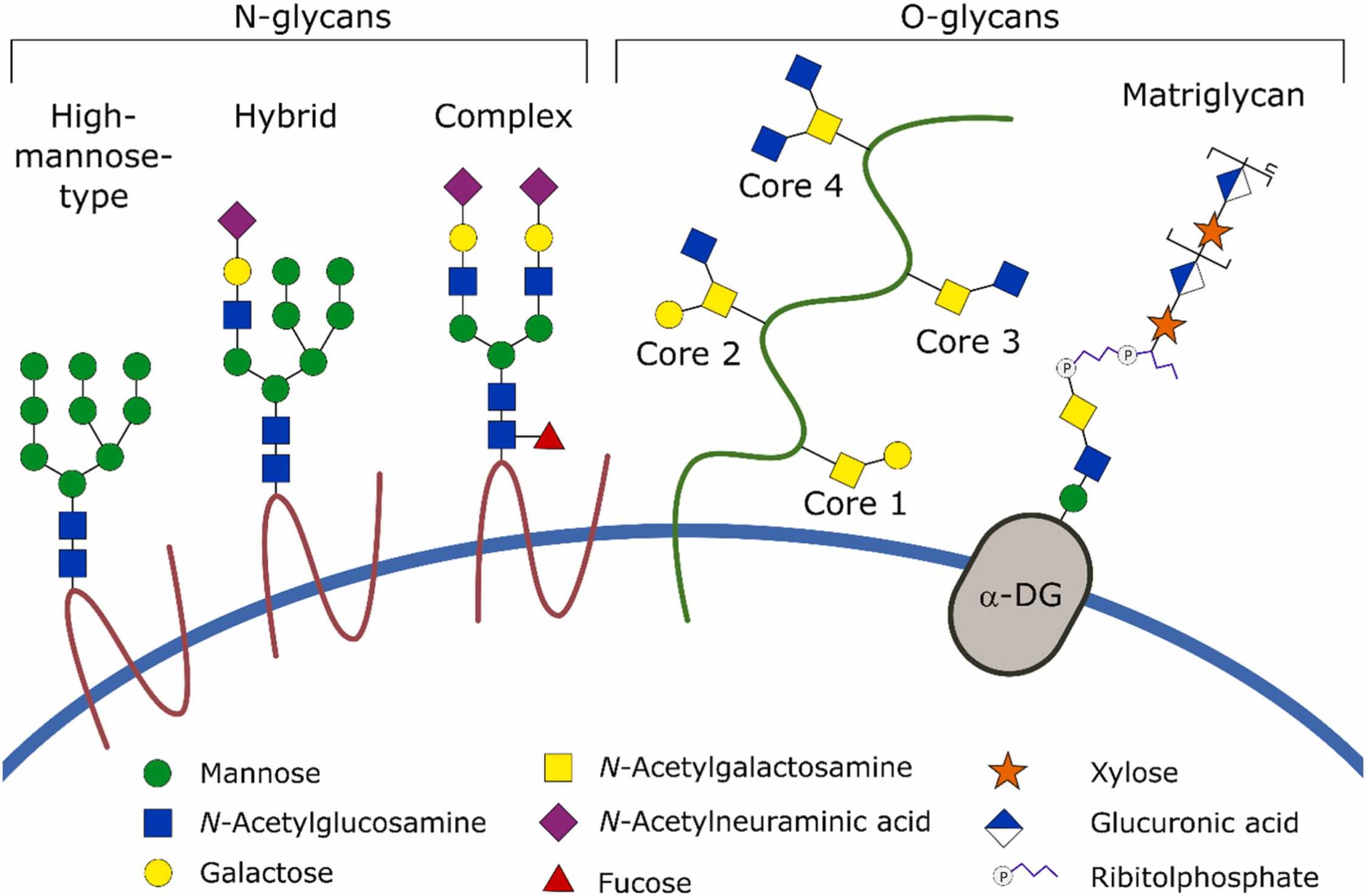
 Figure 1. Common glycan modifications found on mammalian proteins. (Gorzkiewicz et al., 2023)
Figure 1. Common glycan modifications found on mammalian proteins. (Gorzkiewicz et al., 2023)
Profacgen's Glycosylation Profiling service delivers comprehensive identification, quantification, and structural elucidation of protein-attached carbohydrate moieties, providing the analytical foundation required to ensure therapeutic efficacy, regulatory compliance, and manufacturing consistency for glycoprotein biologics.
Glycosylation is among the most structurally complex and functionally consequential post-translational modifications. For monoclonal antibodies, glycan composition directly modulates effector functions such as antibody-dependent cellular cytotoxicity (ADCC) and complement-dependent cytotoxicity (CDC), influences serum half-life through FcRn interactions, and affects immunogenic potential. For recombinant proteins and vaccines, glycosylation governs folding efficiency, stability, and receptor binding. Rigorous glycosylation profiling is therefore essential for clone selection, process development, biosimilar comparability, and regulatory submission.
Protein glycosylation encompasses N-linked glycans attached to asparagine residues within the consensus sequence NXS/T, and O-linked glycans attached to serine or threonine residues. These glycans exhibit remarkable microheterogeneity—varying in antennarity, galactosylation, sialylation, fucosylation, and core structure—that arises from cell line-specific glycosylation machinery, culture conditions, and downstream processing. Regulatory agencies recognize glycosylation as a critical quality attribute (CQA) and require detailed characterization for IND, BLA, and biosimilar applications.
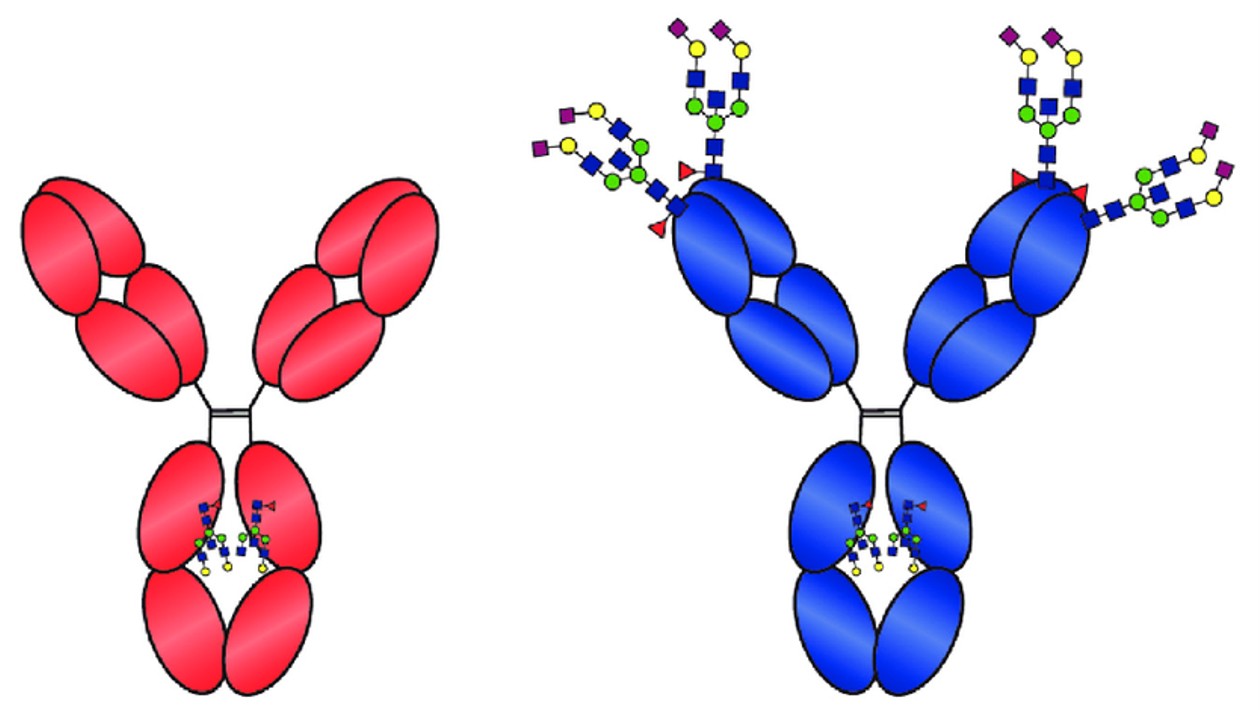 Figure 1. All IgG1 antibodies contain N-linked glycans in the Fc region of the molecule because of the presence of an N-linked glycosylation in the Fc-tail (red antibody). (Steiner and Toes, 2024)
Figure 1. All IgG1 antibodies contain N-linked glycans in the Fc region of the molecule because of the presence of an N-linked glycosylation in the Fc-tail (red antibody). (Steiner and Toes, 2024)
Profacgen's glycosylation platform addresses these requirements through orthogonal analytical techniques that release, separate, identify, and quantify glycan structures with high resolution and reproducibility. Our integrated approach combines chromatographic separation, mass spectrometry, and enzymatic sequencing to deliver unambiguous glycan profiles suitable for regulatory filing, process understanding, and comparability assessment.
These analyses establish a comprehensive glycosylation fingerprint that supports clone selection, formulation development, and regulatory submission.
Our Glycosylation Profiling platform integrates orthogonal analytical methodologies to deliver definitive, stage-appropriate glycan evidence. We tailor technique selection and reporting depth to your regulatory pathway, from early clone screening through formal release testing, biosimilar comparability, and stability monitoring.
Released Glycan Profiling & Quantification
We release and profile glycans to quantify the relative abundance of glycoforms and detect batch-to-batch variation or process drift.
Released glycan profiles provide a global assessment of glycosylation heterogeneity and support lot release and stability protocols.
Site-Specific Glycan Analysis
We determine glycan occupancy and microheterogeneity at individual glycosylation sites to confirm correct post-translational processing and assess site-specific consistency.
This module ensures that glycosylation occurs at the intended residues with the expected heterogeneity.
Glycan Structure Elucidation
We determine the detailed structure of glycans, including linkage positions, antennarity, and branching patterns, to support mechanistic understanding and regulatory documentation.
Structural elucidation data support process understanding, biosimilar analytical similarity, and innovator product characterization.
Glycosylation Comparability & Biosimilar Assessment
We rigorously compare glycosylation profiles across products, batches, or process conditions to demonstrate equivalence or identify meaningful differences.
Comparability protocols are designed to meet FDA and EMA expectations for biosimilar glycosylation assessment.

Program Context:
A biosimilar development team required comprehensive glycosylation comparability data to demonstrate analytical similarity between their candidate monoclonal antibody and the reference product. Regulatory expectations extended beyond profile matching to include correlation with ADCC activity, given the known impact of afucosylation on effector function.
Objective:
To generate a definitive glycosylation comparability package, including released glycan profiling, site-specific analysis, and afucosylated species quantification, with correlation to biological activity data.
Approach:
Profacgen performed N-glycan release with 2-AB labeling, followed by HILIC-UPLC profiling and CE-LIF confirmation for both the biosimilar and reference product. Site-specific glycan occupancy was determined by LC-MS/MS glycopeptide analysis of the Fc N-glycosylation site. Afucosylated glycan content was quantified and compared against the ADCC reporter assay results provided by the client.
Outcome:
The biosimilar demonstrated a glycan profile statistically equivalent to the reference, with afucosylated species within the predefined biosimilarity margin. Site-specific occupancy was comparable, and the afucosylation level correlated with equivalent ADCC activity. The integrated glycosylation and potency package supported successful regulatory submission and progression to clinical comparability studies.
Program Context:
An emerging biotechnology company developing an antibody for oncology indications sought to maximize ADCC activity through glycoengineering. The team needed to screen multiple cell line clones and identify candidates with elevated afucosylation while maintaining acceptable galactosylation and sialylation profiles for acceptable pharmacokinetics and immunogenicity.
Objective:
To profile glycosylation across candidate clones, quantify afucosylated, galactosylated, and sialylated species, and recommend the clone with the optimal balance of effector function and developability.
Approach:
We subjected culture supernatants from six candidate clones to HILIC-UPLC released glycan profiling, quantifying G0, G1, G2, afucosylated, galactosylated, and sialylated glycans. Site-specific occupancy was confirmed by LC-MS/MS, and high-mannose content was monitored as an indicator of incomplete processing. Data were evaluated against predefined target ranges for each glycan species.
Outcome:
One clone demonstrated an afucosylation level 35% higher than the parental line while maintaining galactosylation and sialylation within target ranges and low high-mannose content. This clone was selected for process development, and its glycosylation profile remained stable through scale-up, supporting progression to preclinical efficacy studies with enhanced ADCC potential.
Reference:
Fill out this form and one of our experts will respond to you within one business day.