Charge heterogeneity is a defining characteristic of recombinant protein therapeutics, arising from enzymatic and non-enzymatic post-translational modifications that alter the surface charge distribution of the molecule. Profacgen's Charge Variant & Isoform Analysis services integrate high-resolution separation platforms—including cation-exchange chromatography (CEX), imaged capillary isoelectric focusing (icIEF), and capillary zone electrophoresis (CZE)—with mass spectrometry–based identification to deliver quantitative charge profiling and site-specific modification assignment. Whether confirming batch-to-batch consistency, supporting biosimilar analytical similarity, or investigating stability-related charge drift, our orthogonal approach provides the resolution, sensitivity, and regulatory documentation required for biopharmaceutical quality assurance.
Recombinant monoclonal antibodies and other protein therapeutics are inherently heterogeneous. During cell culture, purification, and storage, a variety of post-translational modifications (PTMs) introduce charge variants that can affect potency, stability, pharmacokinetics, and immunogenicity. According to ICH Q6B, charge heterogeneity must be characterized and monitored as part of the specification for identity, purity, and consistency of biotechnological products.
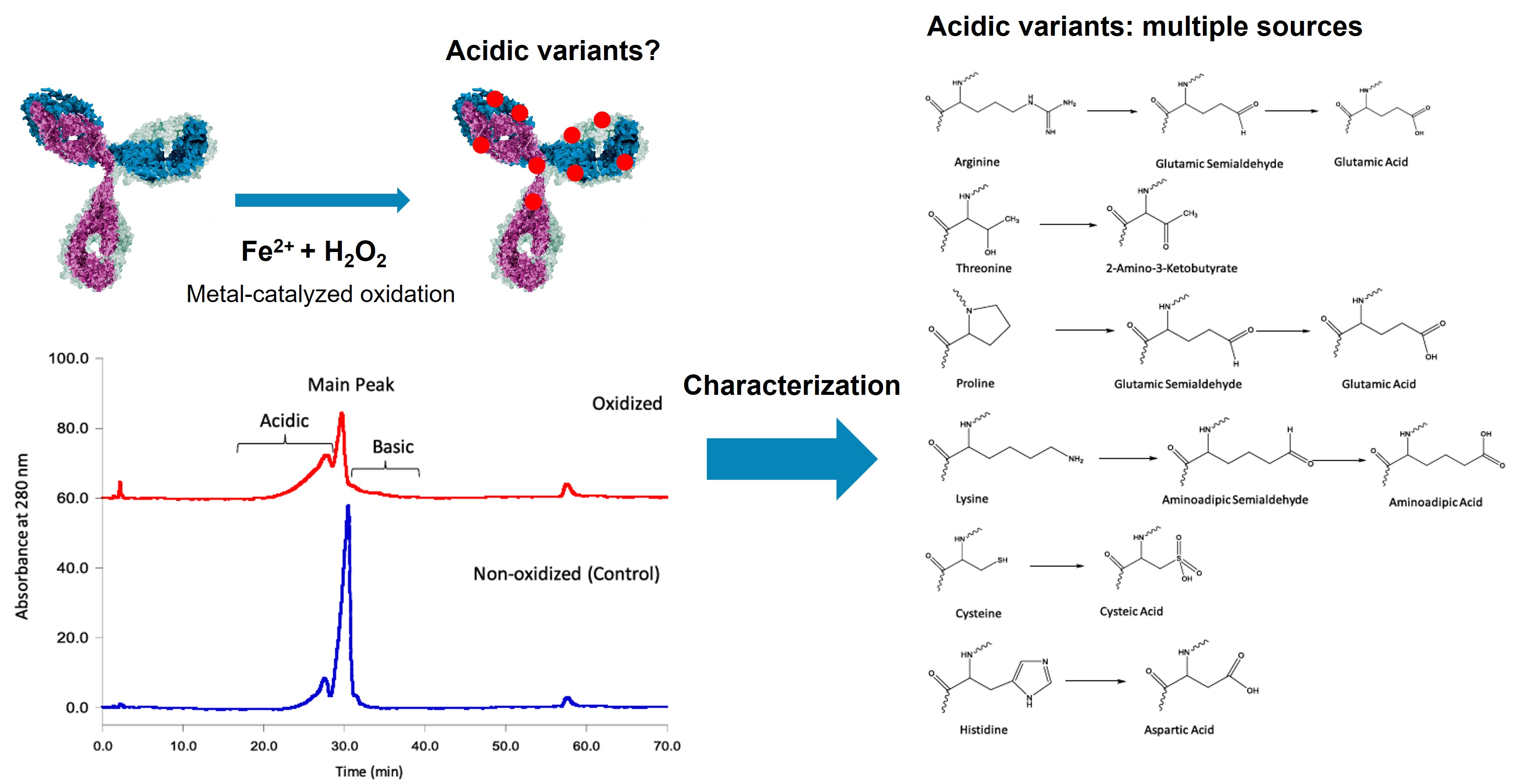 Figure 1. Characterization of acidic variants induced by metal-catalyzed oxidation in a recombinant monoclonal antibody. (Yang et al., 2023)
Figure 1. Characterization of acidic variants induced by metal-catalyzed oxidation in a recombinant monoclonal antibody. (Yang et al., 2023)
The most prevalent charge-altering modifications include deamidation of asparagine and glutamine residues (which introduces a negative charge and shifts pI downward), C-terminal lysine clipping (which removes positive charges and generates acidic species relative to the lysine-retained basic forms), glycation by reducing sugars during cell culture (adding acidic carbonyl groups), sialylation of N- and O-linked glycans (introducing negatively charged sialic acids), and N-terminal pyroglutamylation (removing a positive charge from the N-terminus). These modifications can occur in complementarity-determining regions (CDRs), where even subtle charge changes may impair antigen binding, or in the Fc domain, where they can alter effector function and serum half-life.
Profacgen addresses these analytical demands through a complementary multi-platform strategy. Cation-exchange chromatography (CEX-HPLC) separates variants based on net surface charge under mild nondenaturing conditions, providing robust quantitation for routine release testing. Imaged capillary isoelectric focusing (icIEF) achieves higher resolution by separating proteins according to their isoelectric point (pI), often resolving variants that differ by as little as 0.02 pH units. For direct modification identification, we offer CEX-MS, icIEF-MS, and CZE-MS coupling using volatile buffer systems, as well as fraction collection followed by peptide mapping to pinpoint the exact residues responsible for charge shifts.
Profacgen provides comprehensive charge heterogeneity characterization solutions tailored to research, development, and quality control applications. Our offerings include:
| Service Component | Description |
|---|---|
| Cation-Exchange Chromatography (CEX-HPLC) |
|
| Imaged Capillary Isoelectric Focusing (icIEF) |
|
| Capillary Zone Electrophoresis (CZE) |
|
| Charge Variant Identification by MS |
|

Background:
A biosimilar developer needed to demonstrate analytical similarity between their candidate molecule and the reference product. Charge heterogeneity was identified as a high-risk attribute due to the presence of acidic and basic variants arising from common post-translational modifications. The client required orthogonal data from both cation-exchange chromatography (CEX) and capillary isoelectric focusing (icIEF) to support their comparability assessment.
Our Solution:
Profacgen designed a dual-platform comparability package. CEX-HPLC was performed on a strong cation-exchange column with a linear salt gradient (20–300 mM NaCl in 50 mM sodium phosphate, pH 6.0). icIEF was conducted using a pH 3–10 ampholyte gradient with pI 7.0 and pI 9.0 markers. Both the biosimilar candidate and 12 US reference lots were analyzed in triplicate. Peak areas for acidic variants, main peak, and basic variants were quantified, and statistical equivalence testing was applied to assess similarity.
Final Results:
CEX-HPLC showed acidic variants of 22.4 %, main peak of 72.1 %, and basic variants of 5.5 % for the biosimilar—values that fell within the 95 % confidence interval of the reference lot distribution (acidic: 20.8–24.1 %; main: 70.5–73.8 %; basic: 4.8–6.2 %). icIEF confirmed these results with a main peak pI of 8.42 ± 0.03, matching the reference (8.41 ± 0.04). No new charge species were detected in either platform. The data provided robust evidence of charge profile comparability, supporting the client’s ongoing clinical development.
Background:
A biopharmaceutical company observed a progressive increase in acidic charge variants during real-time stability testing of their Phase III monoclonal antibody at 5 °C. Over 24 months, the acidic fraction increased from 8.2 % to 14.6 %, approaching the release specification limit of 15.0 %. The client needed to identify the specific modification driving the drift, assess its impact on potency, and determine whether the shelf-life claim could be maintained.
Our Solution:
Profacgen employed a tiered analytical strategy. First, icIEF confirmed that the drift was confined to a single acidic species focusing at pI 8.15 (main peak pI 8.42). The acidic species was enriched by preparative weak cation-exchange chromatography (WCX) to >85 % purity. Intact mass analysis of the isolated fraction revealed a +0.984 Da mass shift relative to the main peak, consistent with deamidation. Subunit peptide mapping with AspN digestion localized the modification to Asn52 in the heavy-chain complementarity-determining region 2 (CDR-H2). A cell-based potency assay on the isolated acidic fraction showed a 12 % reduction in target binding affinity compared to the main peak.
Final Results:
The data established that Asn52 deamidation in CDR-H2 was the primary driver of acidic charge drift and the modest potency loss. Because the 24-month level (14.6 %) remained below the 15.0 % specification and the potency impact was within the established clinical exposure range, the client retained the 24-month shelf life with a tightened storage temperature limit (2–8 °C, no excursions above 15 °C). Profacgen's characterization report supported a successful regulatory filing amendment.
Consult Our Experts on Your Project
References:
Fill out this form and one of our experts will respond to you within one business day.