Profacgen's Higher-Order Structure (HOS) Characterization service provides comprehensive biophysical assessment of protein conformation, folding, stability, and assembly state, delivering the structural evidence required to demonstrate functional integrity, manufacturing consistency, and regulatory compliance for biologic therapeutics.
Higher-order structure—encompassing secondary, tertiary, and quaternary organization—directly dictates biological activity, pharmacokinetic behavior, immunogenic potential, and shelf-life stability. Unlike the genetically encoded primary sequence, HOS emerges from complex folding dynamics and environmental interactions, making it exquisitely sensitive to process conditions, formulation composition, and storage history. Rigorous HOS characterization is therefore essential for clone selection, process development, formulation optimization, and comparability assessment.
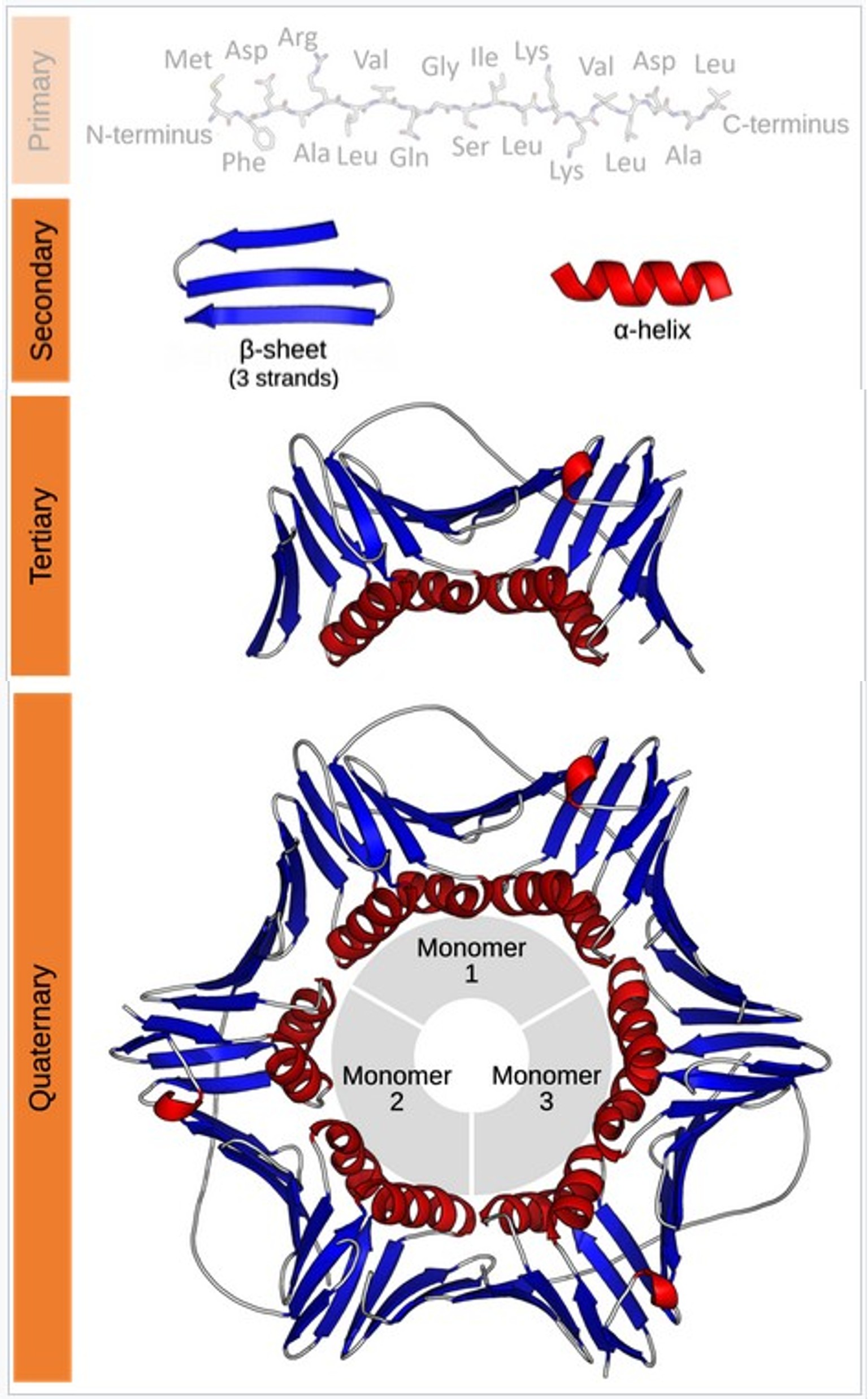 Figure 1. Protein structure hierarchy.
Figure 1. Protein structure hierarchy.
Protein therapeutic function depends not merely on correct amino acid sequence, but on precise three-dimensional folding that positions active sites, maintains stability, and minimizes immunogenic epitopes. Regulatory agencies, including the FDA and EMA, recognize HOS as a critical quality attribute (CQA) and require evidence that the product maintains its intended conformation across manufacturing scales, production batches, and proposed shelf life.
Changes in secondary structure content, tertiary fold integrity, or quaternary assembly state can alter receptor binding affinity, expose cryptic immunogenic epitopes, or accelerate chemical degradation. Profacgen's HOS platform interrogates structural organization at multiple resolutions using orthogonal biophysical techniques that complement one another and eliminate method-specific blind spots, ensuring no conformational change goes undetected:
These analyses establish the structural fingerprint of your biologic and enable sensitive detection of conformational drift induced by process modifications, formulation changes, or forced degradation.
Our HOS Characterization platform integrates orthogonal biophysical methodologies to deliver definitive, stage-appropriate structural evidence. We tailor technique selection, experimental design, and reporting depth to align with your regulatory pathway—from early candidate screening through formal release testing, stability protocols, and biosimilar comparability studies.
Secondary Structure Characterization
Comprehensive assessment of backbone conformation by orthogonal spectroscopic techniques, delivering quantitative secondary structure content for comparability, release testing, and regulatory submission.
Tertiary & Quaternary Structure Characterization
Integrated evaluation of three-dimensional folding, domain integrity, and oligomeric assembly using fluorescence, calorimetry, and solution biophysics to confirm structural fidelity and detect aberrant conformations.
Thermal & Colloidal Stability Assessment
We define the thermal and colloidal stability landscape of your product to guide formulation development, establish storage conditions, and support shelf-life claims.
Stability profiles inform robust formulation selection and identify conditions that preserve native structure.
Aggregation & Quaternary State Analysis
We monitor oligomeric assembly, reversible self-association, and irreversible aggregation that impact potency, safety, and manufacturability.
These methods distinguish native oligomers from pathological aggregates and quantify their abundance.
Program Context:
A biosimilar development team required comprehensive higher-order structure comparability data to demonstrate analytical similarity between their candidate and the reference monoclonal antibody. Regulatory expectations mandated orthogonal evidence at the secondary, tertiary, and thermal stability levels to support the analytical similarity claim and justify reduced clinical immunogenicity assessment.
Objective:
To generate a definitive HOS comparability package, including secondary structure by CD and FTIR, tertiary fold by fluorescence, and thermal stability by DSC, performed side-by-side under identical conditions with statistical evaluation of equivalence.
Approach:
Profacgen executed an orthogonal HOS characterization protocol on both the biosimilar and reference product. Far-UV CD spectra were acquired and deconvoluted for secondary structure content; FTIR amide I bands were analyzed by second-derivative peak fitting; intrinsic fluorescence spectra assessed tryptophan environment; and DSC thermograms were compared for Tm, onset temperature, and unfolding enthalpy.
Outcome:
The biosimilar demonstrated highly similar secondary structure content, equivalent tertiary fold signatures, and superimposable DSC thermograms with matching Tm values. Statistical analysis confirmed analytical similarity across all HOS attributes. The data package supported successful progression to the clinical comparability phase and satisfied regulatory reviewers.
Program Context:
An emerging biotechnology company observed unexpected subvisible particle formation and potency loss in one candidate formulation buffer during early stability studies. The team suspected a formulation-induced conformational change but lacked the biophysical data to identify the structural mechanism or select an optimal buffer.
Objective:
To identify the specific HOS alteration driving aggregation and to guide reformulation by identifying conditions that preserve native secondary structure, tertiary fold, and thermal stability.
Approach:
We subjected the antibody to HOS profiling across four formulation candidates using far-UV CD for secondary structure, intrinsic fluorescence for tertiary integrity, DSC for thermal stability, and DLS for colloidal behavior. Forced degradation at elevated temperature was monitored by SEC-MALS to detect aggregate growth kinetics.
Outcome:
CD and fluorescence revealed partial unfolding and increased solvent exposure of hydrophobic regions in the problematic buffer. DSC showed a reduced Tm and broadened transition, indicating destabilization. A reformulated buffer identified through this screening restored native HOS signatures, increased Tm by 4.2°C, and eliminated particle formation during accelerated stability testing.
Fill out this form and one of our experts will respond to you within one business day.