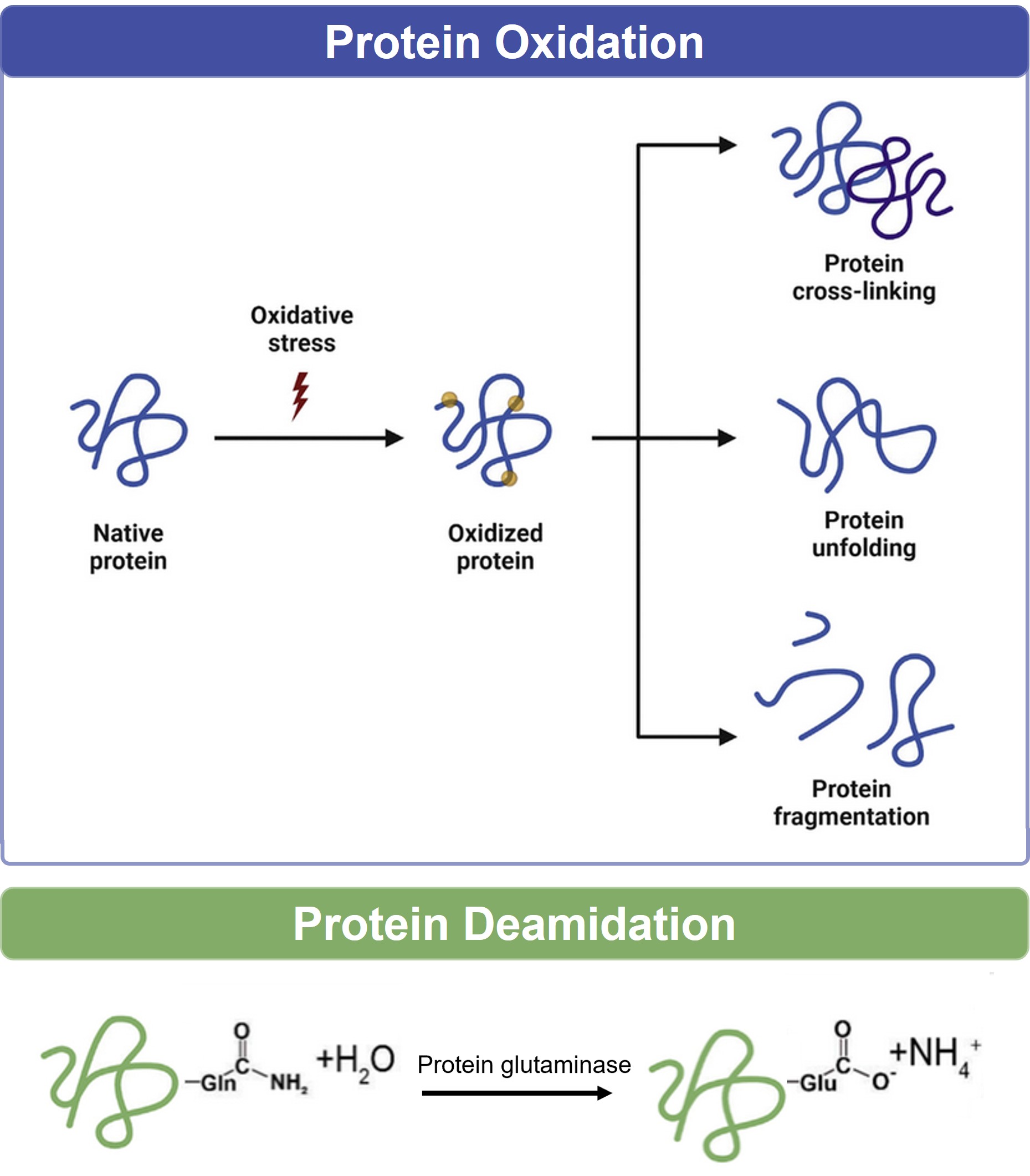
Profacgen's Oxidation & Deamidation Analysis service provides precise identification, site-specific localization, and quantitative assessment of chemical degradation products in protein therapeutics, delivering the data required to ensure product stability, support shelf-life claims, and satisfy regulatory expectations for product-related impurity characterization.
Oxidation and deamidation represent two of the most prevalent chemical degradation pathways in biologics. Methionine and tryptophan oxidation, asparagine deamidation, and aspartate isomerization can reduce potency, alter pharmacokinetics, increase immunogenicity risk, and compromise shelf-life stability. Rigorous analytical characterization of these modifications is therefore essential for defining critical quality attributes, setting release specifications, and supporting IND, BLA, and biosimilar submissions.
Chemical degradation of protein therapeutics occurs through reactions with reactive oxygen species, hydrolytic cleavage, and pH-dependent rearrangements. Methionine residues are particularly susceptible to oxidation by peroxides and dissolved oxygen, while asparagine residues undergo base-catalyzed deamidation via a cyclic imide intermediate that can isomerize to isoaspartate. These modifications are not merely theoretical liabilities—they are frequently observed during manufacturing, storage, and stress conditions, and regulatory agencies require their quantification as product-related substances.
Profacgen's platform addresses these requirements through orthogonal analytical techniques that localize modifications to specific residues, quantify their abundance relative to the unmodified species, and assess their rate of formation under accelerated conditions. Our integrated approach combines peptide mapping, charge-based separations, and forced degradation studies to deliver comprehensive, regulatory-compliant data packages.
These analyses establish quantitative baselines for product-related impurities and inform formulation selection, process control, and acceptance criteria.
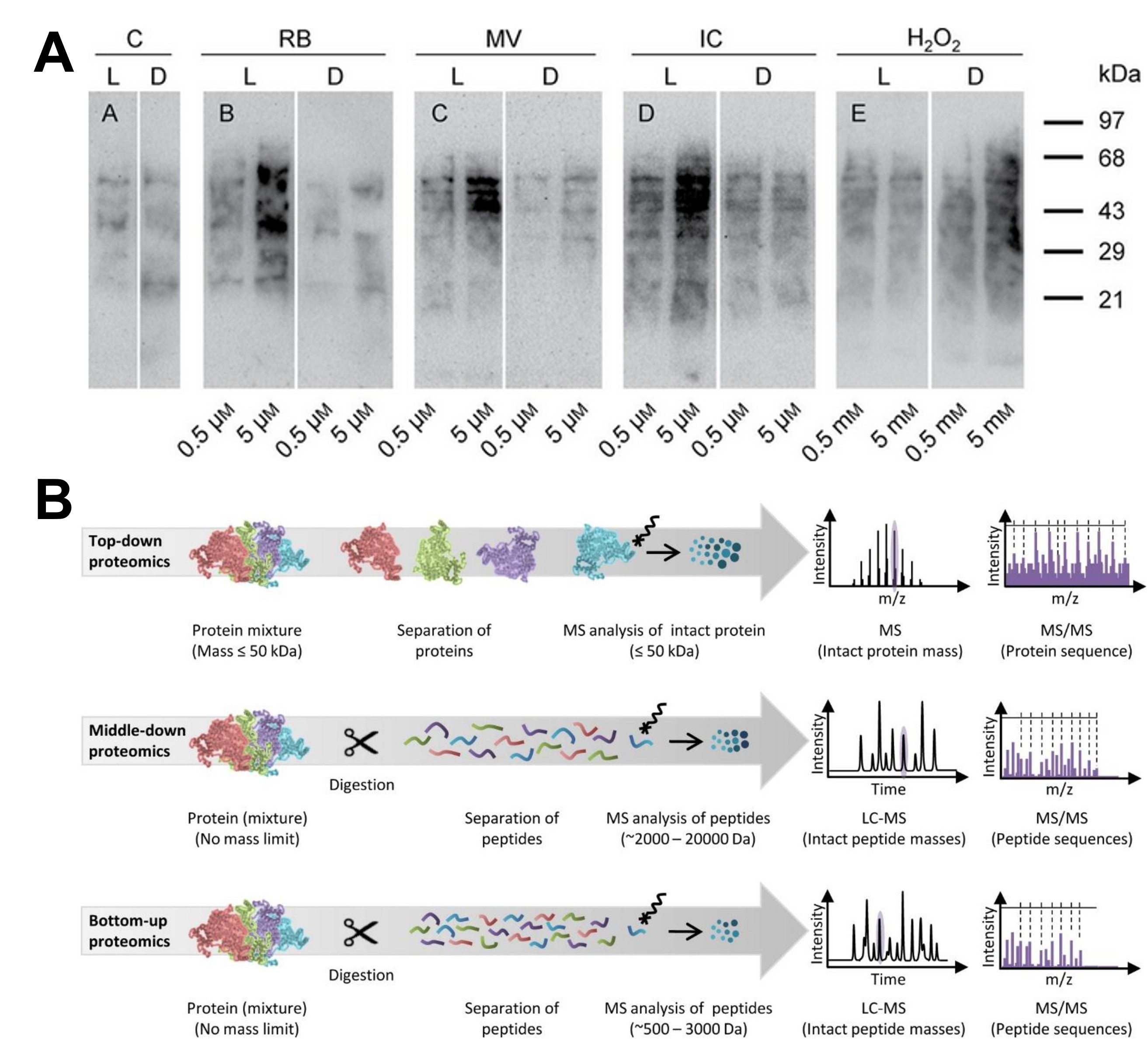 Figure 1. Examples of protein oxidation and deamidation analysis. (A) Protein oxidation analysis OxyBlot of ACSC under (photo)oxidative stress conditions. (Gutiérrez et al., 2014) (B) Mass spectrometric analysis of protein deamidation. (Jin et al., 2022)
Figure 1. Examples of protein oxidation and deamidation analysis. (A) Protein oxidation analysis OxyBlot of ACSC under (photo)oxidative stress conditions. (Gutiérrez et al., 2014) (B) Mass spectrometric analysis of protein deamidation. (Jin et al., 2022)
Our Oxidation & Deamidation Analysis platform integrates orthogonal analytical methodologies to deliver definitive, stage-appropriate degradation evidence. We tailor technique selection and reporting depth to your regulatory pathway, from early liability assessment through formal release testing, stability protocols, and biosimilar comparability studies.
Methionine & Tryptophan Oxidation Analysis
We detect and quantify oxidation at labile residues to assess process-induced and storage-related degradation, supporting specification setting and root-cause investigation.
Oxidation profiling identifies sequence hotspots and informs antioxidant formulation strategies.
Asparagine Deamidation & Aspartate Isomerization
We characterize the most common degradation pathway in protein therapeutics, quantifying deamidation and isomerization rates to support shelf-life claims and process optimization.
This module directly supports stability protocol design and acceptance criteria justification.
Glutamine Deamidation & Chemical Stress Profiling
We assess glutamine deamidation and broader chemical degradation under forced conditions to identify liabilities and validate analytical methods for stability monitoring.
Stress profiling accelerates mechanistic understanding and validates stability-indicating methods.
Quantification, Specification & Forced Degradation
We provide quantitative data and kinetic modeling to support specification setting, shelf-life projection, and regulatory filing for product-related impurities.
This module transforms analytical data into actionable quality parameters and regulatory documentation.

Program Context:
A biosimilar development team required comprehensive oxidation and deamidation comparability data to demonstrate analytical similarity between their candidate monoclonal antibody and the reference product. Regulatory expectations mandated site-specific quantification of degradation products with predefined equivalence margins.
Objective:
To generate a definitive degradation product comparability package, including methionine oxidation, asparagine deamidation, and aspartate isomerization quantification, performed side-by-side under identical conditions with statistical evaluation.
Approach:
Profacgen performed LC-MS/MS peptide mapping with multiple protease digestions on both the biosimilar and reference product. Oxidized methionine-containing peptides and deamidated asparagine-containing peptides were identified by characteristic mass shifts, quantified as relative percentages, and compared using equivalence margin testing.
Outcome:
The biosimilar demonstrated oxidation and deamidation levels statistically equivalent to the reference at all monitored residues, with no degradation hotspots unique to either product. The data package supported successful regulatory submission and progression to clinical comparability studies.
Program Context:
A pharmaceutical company observed progressive potency decline and increased acidic charge variants during long-term stability studies of a therapeutic antibody. The team suspected chemical degradation but required identification of the specific modification and its rate of formation to guide reformulation.
Objective:
To identify and localize the chemical degradation responsible for potency loss, quantify its formation kinetics, and recommend formulation modifications to mitigate the degradation pathway.
Approach:
We subjected stressed and control samples to icIEF for charge variant quantification, then performed LC-MS/MS peptide mapping to localize modifications. IEX fractionation isolated acidic species for targeted analysis, and forced degradation studies at varied pH and temperature identified the optimal formulation window.
Outcome:
Peptide mapping identified a complementarity-determining region asparagine residue undergoing rapid deamidation with isoaspartate formation, directly correlating with the potency decline. A reformulated buffer at reduced pH decreased the deamidation rate by 55% during accelerated testing, supporting an extended shelf-life claim and revised release specifications.
References:
Fill out this form and one of our experts will respond to you within one business day.